-
山茶花(Camellias)是我国传统名花,花色为其重要观赏性状[1]。花青苷为山茶花花色形成的物质基础,总花青苷及主要花青苷含量决定其花色[2-4]。研究还表明,花青苷具有抑制肿瘤[5-6]、降血糖[7]和抗氧化[8-9]等作用,可用于医药、保健和化妆等产品的开发。Li等[10-11]利用核磁共振、质谱和紫外可见光谱等技术研究了山茶花花青苷,分别在滇山茶(Camellia reticulata Lindl.)中鉴定出5种含2-O-β-木糖基及其相应不含2-O-β-木糖基的花青苷,滇山茶园艺品种‘大理茶’中鉴定出10种含2-O-β-木糖基花青苷。李辛雷等[2,4]应用高效液相色谱-光电二极管阵列检测和超高效液相色谱-四极杆-飞行时间质谱联用技术,从山茶(C. japonica L.)不同花色品种中鉴定了出7种花青苷,且其花色随总花青苷及主要花青苷含量增加而加深。
杜鹃红山茶(C. azalea Wei)四季开花,是培育四季茶花的优良亲本[12],广州棕科园艺开发有限公司通过杜鹃红山茶与山茶‘媚丽’杂交,已培育出新品种50余个[13]。已有杜鹃红山茶研究主要集中于种群结构 [14]、生理特性[15]、生物学特性[16]和繁殖[17-18]等方面,李辛雷等[19]对其色素成分进行了初步研究,但其花青苷从亲本到杂交后代的变异特征尚不清楚。鉴于此,本试验应用高效液相色谱-光电二极管阵列检测(HPLC-DAD)和超高效液相色谱-四极杆-飞行时间质谱联用技术(UPLC-Q-TOF-MS),对杜鹃红山茶、山茶‘媚丽’及其杂交后代花青苷成分与含量进行研究,明确其花青苷变异特征,以期为高花青苷含量山茶花新品种选育提供科学依据,进一步为其花青苷功能产品的开发利用提供物质基础。
-
试验材料为杜鹃红山茶、山茶‘媚丽’及其35个杂交F1代。杜鹃红山茶和山茶‘媚丽’分别标记为D和M,以山茶‘媚丽’为母本,杜鹃红山茶为父本的27个杂交后代标记为MD系列,以杜鹃红山茶为母本,山茶‘媚丽’为父本的8个杂交后代标记为DM系列。杜鹃红山茶花色为鲜红色,单瓣型,山茶‘媚丽’花色为玫瑰红色,花瓣边缘带有白边,半重瓣型至牡丹型。杜鹃红山茶与山茶‘媚丽’杂交后代花色深红至浅红不等,花型有单瓣型、半重瓣型和托桂型等(图1)。所有材料采自广州棕科园艺开发有限公司。选取生长环境相同,长势一致的植株5株,对盛开期的花朵进行随机采样,每个植株采3朵,装入密封袋后置于有保鲜冰袋的保鲜箱中保存。样品带回实验室后,−80 ℃冰箱中保存。
-
使用液氮研磨花瓣,称取2 g倒入试管中,加入5 mL提取液(三氟醋酸:甲酸:水:甲醇,体积比1:2:27:70),摇匀,封口膜封口,常温下浸提24 h。浸提结束后,经脱脂棉和0.22 μm的滤膜依次过滤,滤液倒入2 mL棕色进样瓶中密封保存,最后置于−20 ℃冰箱中备用[20-21]。
应用Waters(Waters Alliance 2695,USA)的高效液相色谱-光电二极管阵列检测(HPLC-DAD)分析系统进行色素分析。色谱柱为TSK gel ODS-100Z(150 mm × 4.6 mm ,柱内直径为5 μm,Tosoh,Japan)。分析条件:柱温40 ℃,流速1 mL·min−1,进样量20 μL,检测波长525 nm。流动相A为水:磷酸(体积比98.5:1.5);流动相B为水:乙腈:甲酸:四氢呋喃:磷酸(体积比48.5:25:20:5:1.5),配制的溶液在减压条件下用0.22 μm的微孔滤膜过滤,超声脱气后备用。梯度洗脱程序:0~15 min,B液浓度从22%上升至28%;15~35 min,B液从28%上升至68%[22]。
利用超高效液相色谱-四极杆-飞行时间质谱联用技术(UPLC-Q-TOF-MS),结合混合标准品Cy3Ga和Cy3G共洗脱试验,进行花青苷结构鉴定。设备为ACQUITYTM UPLCⅠ-Class 超高效液相色谱系统(Waters Corporation,Milford,MA,USA),Xevo G2-XS QTof MS质谱系统(Waters Corporation,Manchester,UK),UNIFI 1.8软件系统。色谱条件与上述色素定性分析条件一致。质谱分析条件:电喷雾电离离子源(ESI),正离子模式,全离子扫描,扫描范围(m/z):50~1200 u。脱溶剂气体为高纯度氮气,温度450 ℃,流速600 L·h−1,毛细管电压为1 kV,锥孔电压为40 V。低能量扫描电压为6 eV,高能量扫描电压为20~45 eV。
-
标准品矢车菊素-3-O-β-葡萄糖苷(Cy3G)和矢车菊素-3-O-β-半乳糖苷(Cy3Ga)购于上海源叶生物科技有限公司(纯度≥98%)。根据朱满兰等[23]方法,采用标准品半定量法计算各样品每克新鲜花瓣中含有的相对于标准品的花青苷含量(μg·g−1),重复3次。
-
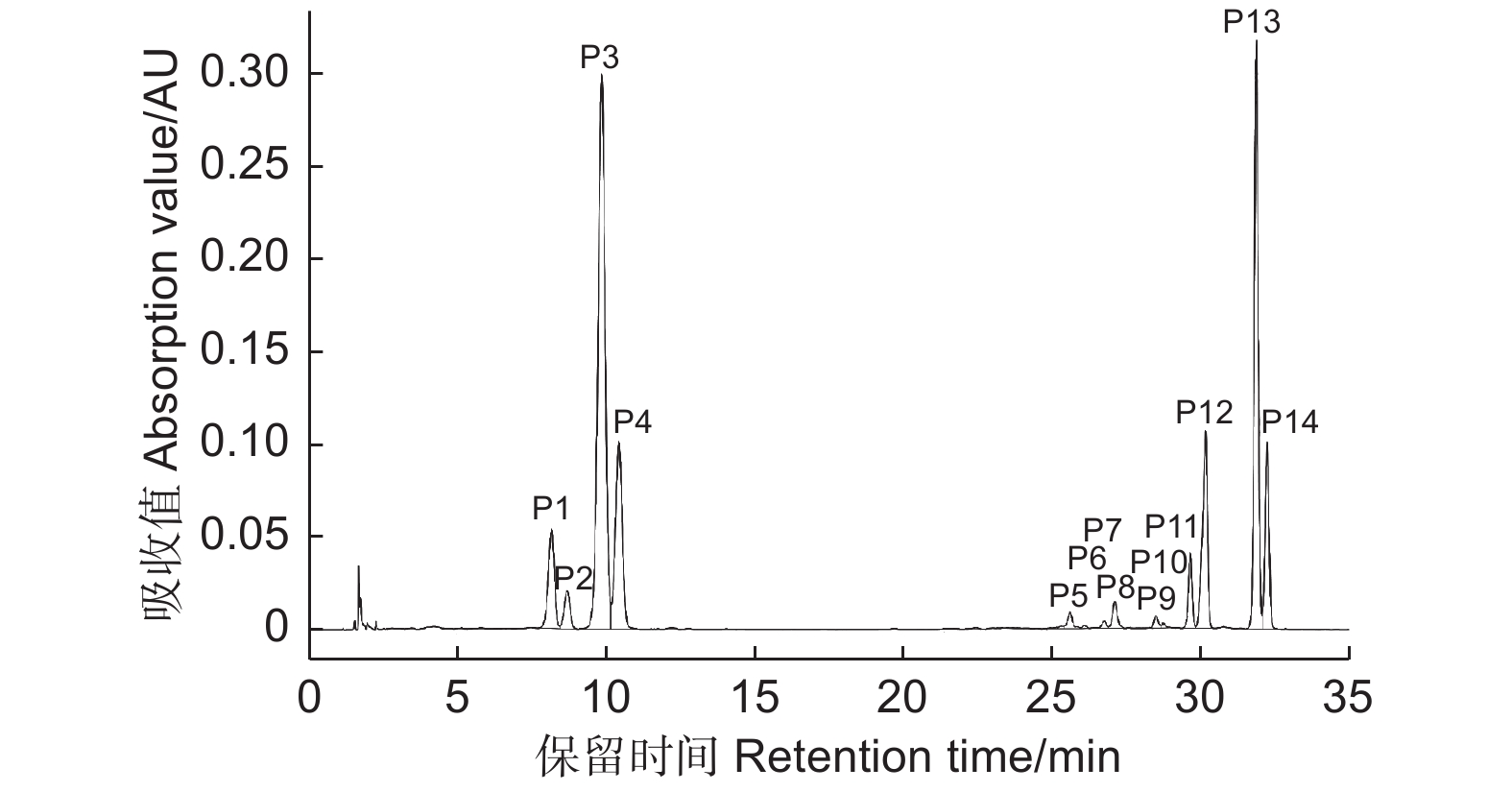
根据UPLC-Q-TOF-MS图谱,对杜鹃红山茶、山茶‘媚丽’及其杂交后代花瓣中花青苷成分进行鉴定,共检测到14种花青苷(图2),其紫外-可见光谱及质谱数据见表1。根据Cy糖苷在513~520 nm有特征吸收峰及碎片离子m/z 287,推定14种花青苷均为Cy型花青苷[24];花青苷在440 nm与λvis-max两处吸收值之比A440/Avis-max变化范围为32%~34%,确定14种花青苷均为3-O-糖苷类型[25-26]。根据290~340 nm波长下肩峰的出现推定化合物被芳香酸酰化[27],峰P5~P14在310~316 nm波长下肩峰的出现推定其花青苷被芳香酸酰化。
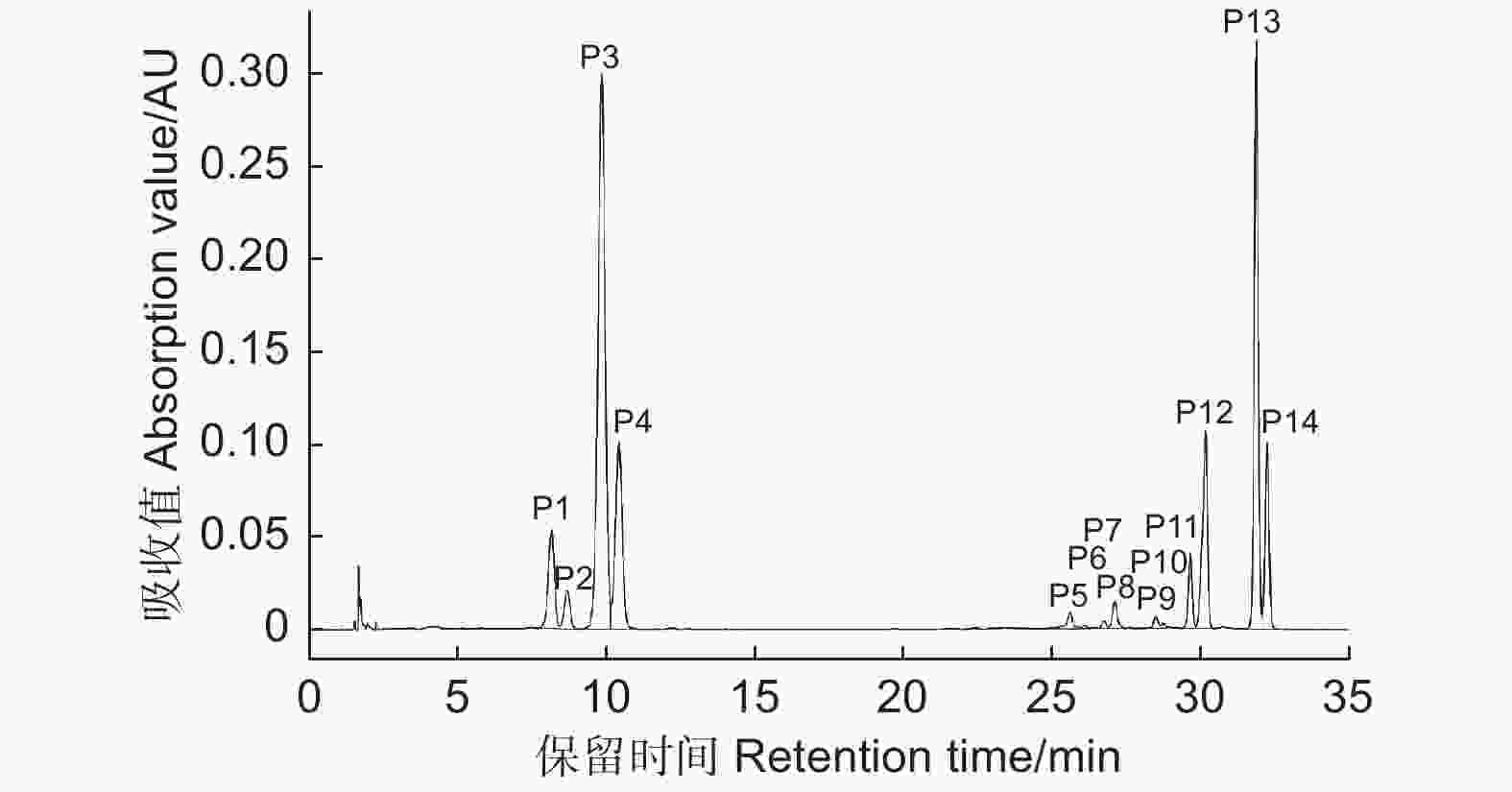
Figure 2. HPLC chromatogram of anthocyanin components in hybrids of C. azalea and C. japonica 'Meili'
色谱峰
Peak
No.保留时间
Retention time
/min吸收波长
λmax
/nmA440/
Avis-max
/%分子离子
Molecular ions
(m/z)碎片离子
Fragment ions
(m/z)推定结果
Tentative
identification参考文献
ReferenceP1 8.25 281,516 32 581 449,287 Cy3GaX [10-11] P2 8.78 282,516 32 449 287 Cy3Ga [2-4] P3 9.98 282,516 33 581 449,287 Cy3GX [10-11] P4 10.54 282,514 32 449 287 Cy3G [2-4] P5 25.64 281,315,516 33 611 449,287 Cy3GaECaf [2-4] P6 25.89 282,315,517 33 743 611,449,287 Cy3GaECafX [10-11] P7 26.76 284,311,515 34 727 595,449,287 Gy3GaZpCX [10-11] P8 27.19 285,310,516 34 595 449,287 Gy3GaZpC [10-11] P9 28.52 283,315,515 33 743 611,449,287 Cy3GECafX [10-11] P10 28.77 283,316,515 33 611 449,287 Cy3GECaf [2-4] P11 29.68 283,313,516 34 595 449,287 Cy3GaEpC [2-4] P12 30.07 282,312,516 33 727 595,449,287 Cy3GaEpCX [10-11] P13 31.89 284,314,514 34 727 595,449,287 Cy3GEpCX [10-11] P14 32.24 283,313,515 34 595 449,287 Cy3GEpC [2-4] 注:Cy3GaX: 矢车菊素-3-O-(2-O-β-木糖基)-β-半乳糖苷; Cy3Ga: 矢车菊素-3-O-β-半乳糖苷; Cy3GX: 矢车菊素-3-O-(2-O-β-木糖基)-β-葡萄糖苷; Cy3G: 矢车菊素-3-O-β-葡萄糖苷; Cy3GaECaf: 矢车菊素-3-O-[6-O-(E)–咖啡酰]-β-半乳糖苷; Cy3GaECafX: 矢车菊素-3-O-[2-O-β-木糖基-6-O-(E)-咖啡酰]-β-半乳糖苷; Cy3GaZpCX: 矢车菊素-3-O-[2-O-β-木糖基-6-O-(Z)-p-香豆酰]-β-半乳糖苷; Cy3GaZpC: 矢车菊素-3-O-[6-O-(Z)-p-香豆酰]-β-半乳糖苷; Cy3GECafX: 矢车菊素-3-O-[2-O-β-木糖基-6-O-(E)-咖啡酰]-β-葡萄糖苷; Cy3GECaf: 矢车菊素-3-O-[6-O-(E)-咖啡酰]-β-葡萄糖苷; Cy3GaEpC: 矢车菊素-3-O-[6-O-(E)-p-香豆酰]-β-半乳糖苷; Cy3GaEpCX: 矢车菊素-3-O-[2-O-β-木糖基-6-O-(E)-p-香豆酰]-β-半乳糖苷; Cy3GEpCX: 矢车菊素-3-O-[2-O-β-木糖基-6-O-(E)-p-香豆酰]-β-葡萄糖苷; Cy3GEpC: 矢车菊素-3-O-[6-O-(E)-p-香豆酰]-β-葡萄糖苷。下同。
Notes: Cy3GaX: Cyanidin-3-O-(2-O-β-xylosyl)-β-galactoside; Cy3Ga: Cyanidin-3-O-β-galactoside; Cy3GX: Cyanidin- 3-O-(2-O-β-xylosyl)-β-glucoside; Cy3G: Cyanidin-3-O-β-glucoside; Cy3GaECaf: Cyanidin- 3-O-(6-O-(E)-caffeoyl)-β-galactoside; Cy3GaECafX: Cyanidin-3-O-(2-O-β-xylosyl-6-O-(E)-caffeoyl)-β-galactoside; Cy3GaZpCX: Cyanidin-3-O-(2-O-β-xylosyl-6-O-(Z)-p-coumaroyl)-β-galactoside; Cy3GaZpC: Cyanidin- 3-O-(6-O-(Z)-p-coumaroyl)-β-galactoside; Cy3GECafX: Cyanidin- 3-O-(2-O-β-xylosyl-6-O-(E)-caffeoyl)-β-glucoside; Cy3GECaf: Cyanidin-3-O-(6-O-(E)-caffeoyl)-β-glucoside; Cy3GaEpC: Cyanidin-3-O-(6-O-(E)-p-coumaroyl)-β-galactoside; Cy3GaEpCX: Cyanidin- 3-O-(2-O-β-xylosyl-6-O-(E)-p-coumaroyl)-β-galactoside; Cy3GEpCX: Cyanidin-3-O-(2-O-β-xylosyl-6-O-(E)-p-coumaroyl)-β-glucoside; Cy3GEpC: Cyanidin- 3-O-(6-O-(E)-p-coumaroyl)-β-glucoside. The same as below.Table 1. Chromatographic and spectral data of anthocyanins in hybrids of C. azalea and C. japonica 'Meili'
峰P2和P4质谱数据为分子离子m/z 449,碎片离子m/z 287,其中,m/z 287为Cy苷元特征质荷比,其裂解释放162 u对应一分子己糖;根据峰P2和峰P4与标准品Cy3Ga和Cy3G共洗脱特性以及花青素半乳糖洗脱时间小于花青素葡萄糖苷特性[24],确定峰P2为矢车菊素-3-O-β-半乳糖苷,峰P4为矢车菊素-3-O-β-葡萄糖苷[28-29]。峰P1和P3质谱数据为分子离子m/z 581,碎片离子m/z 449、287,m/z 581到m/z 449丢失132 u,m/z 449到m/z 287丢失162 u,参考Li等[10-11]文献,推定其分别为矢车菊素-3-O-(2-O-β-木糖基)-β-半乳糖苷(Cy3GaX)、矢车菊素-3-O-(2-O-β-木糖基)-β-葡萄糖苷(Cy3GX)。
峰P5和P10质谱数据为分子离子m/z 611,碎片离子m/z 449、287,m/z 611到m/z 449丢失162 u,m/z 449到m/z 287丢失162 u,判定其为矢车菊素-3-O-[6-O-(E)-咖啡酰]-β-半乳糖苷(Cy3GaECaf)和矢车菊素-3-O-[6-O-(E)-咖啡酰]-β-葡萄糖苷(Cy3GECaf)[2-4]。峰P6和P9质谱数据为分子离子m/z 743,碎片离子m/z 611、449、287,m/z 743到m/z 611丢失132 u,m/z 611到m/z 449丢失162 u,m/z 449到m/z 287丢失162 u,推定其为矢车菊素-3-O-(2-O-β-木糖基-6-O-(E)-咖啡酰)-β-半乳糖苷(Cy3GaECafX)和矢车菊素-3-O-(2-O-β-木糖基-6-O-(E)-咖啡酰)-β-葡萄糖苷(Cy3GECafX)。
峰P8、P11和P14质谱数据为分子离子m/z 595,碎片离子m/z 449、287,m/z 595到m/z 449丢失146 u,m/z 449到m/z 287丢失162 u;根据其A440/Avis-max(34%)及在310、313 nm波长下肩峰的出现,推定其为Cy-3-O-芳香酸酰化型糖苷而排除鼠李糖苷[30];根据顺式花青苷洗脱时间小于反式花青苷[31-32]及花青素半乳糖苷洗脱时间小于花青素葡萄糖苷的特性,判定峰P8、P11和P14分别为矢车菊素-3-O-[6-O-(Z)-p-香豆酰]-β-半乳糖苷(Cy3GaZpC)、矢车菊素-3-O-[6-O-(E)-p-香豆酰]-β-半乳糖苷(Cy3GaEpC)和矢车菊素-3-O-[6-O-(E)-p-香豆酰]-β-葡萄糖苷(Cy3GEpC)。
峰P7、P12和P13质谱数据为分子离子m/z 727,碎片离子m/z 595、449、287,m/z 727到m/z 595丢失132 u,m/z 595到m/z 449丢失146 u,m/z 449到m/z 287丢失162 u,推定其分别为矢车菊素-3-O-[2-O-β-木糖基-6-O-(Z)-p-香豆酰]-β-半乳糖苷(Cy3GaZpCX)、矢车菊素-3-O-[2-O-β-木糖基-6-O-(E)-p-香豆酰]-β-半乳糖苷(Cy3GaEpCX)和矢车菊素-3-O-[2-O-β-木糖基-6-O-(E)-p-香豆酰]-β-葡萄糖苷(Cy3GEpCX)[10-11]。
-
杜鹃红山茶、山茶‘媚丽’及其35个杂交后代中,‘媚丽’检测到7种花青苷,其中Cy3G、Cy3Ga、Cy3GEpC和Cy3GaEpC含量较高,Cy3GaECaf、Cy3GaZpC和Cy3GECaf含量均低于10 μg·g−1。杜鹃红山茶中检测到14种花青苷,除与山茶‘媚丽’相同的7种外,还包括其相应的含2-O-β-木糖基的花青苷;含量较高的花青苷有8种(表2),分别为Cy3G、Cy3Ga、Cy3GEpC和Cy3GaEpC以及相应的含2-O-β-木糖基的Cy3GX、Cy3GaX、Cy3GEpCX和Cy3GaEpCX,其余成分均低于1 μg·g−1。杜鹃红山茶与山茶‘媚丽’35个杂交后代除MD78外,均检测到8种主要相关花青苷,其余成分含量均较低或未检测到。
样品 Samples Cy3GaX Cy3Ga Cy3GX Cy3G Cy3GaEpCX Cy3GaEpC Cy3GEpCX Cy3GEpC 合计 Total M — 28.23±2.15 — 215.43±15.70 — 43.75±2.55 — 239.21±12.68 526.61±30.67 D 141.54±0.47 5.75±0.00 466.23±1.57 44.88±0.16 23.38±0.07 2.07±0.01 40.05±0.12 11.90±0.03 735.81±2.43 MD2 8.38±0.43 1.70±0.02 120.51±0.78 25.76±0.20 24.35±0.74 21.04±0.52 125.89±4.31 34.18±1.42 361.80±7.28 MD4 5.56±0.46 6.81±0.33 85.48±4.70 163.12±9.15 57.52±2.64 10.50±0.47 108.55±5.92 293.89±17.35 731.42±40.29 MD6 17.82±0.10 29.37±0.96 189.85±4.36 561.41±16.24 78.56±2.88 6.62±0.34 136.90±4.33 385.62±11.67 1406.15±40.60 MD10 29.25±0.58 2.54±0.06 392.53±10.24 29.80±0.96 32.91±1.01 20.20±0.52 171.04±5.41 34.88±0.64 713.15±18.34 MD11 35.13±0.80 10.78±0.50 366.20±10.17 105.87±3.84 31.48±1.00 10.96±0.28 115.86±3.46 31.16±0.93 707.45±20.81 MD13 47.85±1.37 6.99±0.30 200.59±6.58 31.68±1.06 47.06±1.92 6.54±0.19 63.09±2.22 21.28±1.20 425.08±14.58 MD14 5.89±0.23 3.81±0.20 61.03±1.81 46.92±28.29 39.04±1.90 6.72±0.25 63.89±2.29 137.81±7.90 365.11±17.43 MD16 27.63±0.67 9.84±0.39 141.62±3.56 64.23±1.86 88.17±3.66 14.15±0.47 132.94±4.70 75.58±2.29 554.15±17.33 MD17 4.53±0.19 3.20±0.20 44.43±0.93 45.16±1.94 46.88±2.01 10.22±0.65 55.05±2.13 151.13±5.51 360.60±10.59 MD19 11.26±0.19 3.78±0.08 213.63±5.02 82.64±2.20 25.56±0.84 19.46±0.58 119.21±4.19 59.73±2.04 535.26±14.87 MD25 71.58±1.22 25.06±0.85 296.79±7.71 144.74±4.26 53.97±1.86 9.52±0.26 62.47±1.88 40.19±1.41 704.32±19.41 MD26 15.67±0.42 5.16±0.21 121.23±2.96 90.56±2.84 26.51±1.04 4.87±0.12 60.60±1.87 76.01±3.90 400.62±12.73 MD28 16.92±0.33 26.95±0.93 117.73±2.28 404.54±11.30 42.36±1.23 5.75±0.17 52.93±1.31 134.92±3.63 802.10±20.82 MD29 75.06±2.68 10.31±0.56 247.46±9.47 52.06±2.27 50.62±2.05 11.12±0.37 79.52±3.00 18.59±0.60 544.74±20.75 MD36 25.58±0.02 5.11±0.13 297.94±5.12 61.06±1.57 51.02±1.07 17.45±0.29 245.78±5.83 42.02±0.62 745.96±13.88 MD51 32.13±0.12 11.11±0.42 202.10±4.49 65.13±2.00 49.53±1.67 14.86±0.39 115.04±3.31 36.94±1.19 526.84±13.49 MD52 20.28±0.86 6.24±0.42 64.94±2.97 30.45±1.95 19.40±0.98 6.73±0.17 38.10±1.64 19.35±1.75 205.49±10.60 MD55 12.72±0.18 8.59±0.38 147.86±4.38 159.45±6.94 33.86±1.43 9.44±0.34 63.87±2.05 91.74±5.41 527.53±20.67 MD58 5.43±0.43 1.38±0.12 148.40±6.29 48.22±2.32 25.57±0.62 5.40±0.18 231.67±6.20 133.23±4.19 599.30±19.03 MD61 13.01±0.48 4.40±0.05 183.65±2.61 64.41±0.41 40.28±0.67 17.82±0.38 139.72±2.68 66.94±0.29 530.23±6.65 MD71 7.69±0.20 0.54±0.06 151.50±5.97 12.13±0.49 12.81±0.32 13.42±0.22 99.70±3.21 11.99±0.53 309.78±10.13 MD72 20.99±0.42 2.50±0.09 403.17±8.23 72.35±1.94 17.82±0.50 17.10±0.30 107.89±2.82 22.98±0.70 664.79±14.82 MD73 8.58±0.33 7.46±0.12 83.90±0.49 122.28±0.99 66.67±1.25 12.96±0.33 113.60±1.51 198.84±2.32 614.28±4.71 MD75 18.34±0.35 16.63±0.73 166.68±4.85 250.27±8.99 42.84±1.63 9.11±0.30 71.60±2.35 126.47±4.01 701.94±23.12 MD76 3.06±0.15 1.62±0.10 66.26±1.13 71.27±1.55 27.10±0.21 7.80±0.05 79.77±0.78 123.34±0.76 380.23±4.41 MD77 122.71±2.05 7.85±0.04 409.23±8.97 42.62±0.62 78.89±2.43 11.22±0.30 97.25±2.64 17.84±0.32 787.61±17.30 MD78 1.29±0.01 — 38.99±0.15 7.36±0.05 6.85±0.03 11.03±0.05 51.73±0.09 17.07±0.03 134.32±0.31 DM3 15.95±0.59 5.24±0.25 196.31±4.31 87.41±2.76 36.88±1.39 17.38±0.56 215.33±7.68 77.81±2.09 652.32±18.26 DM4 7.20±0.11 2.39±0.03 85.17±0.28 28.77±0.06 33.49±1.10 21.63±0.57 149.18±4.98 64.55±1.45 392.38±8.30 DM5 12.10±0.33 3.06±0.12 198.38±6.15 67.83±3.02 32.01±1.47 14.22±0.58 217.57±11.09 61.49±3.53 606.66±25.62 DM7 15.40±0.27 3.66±0.18 174.09±6.82 43.46±2.21 31.28±1.54 11.85±0.48 160.62±7.20 27.73±0.89 468.08±18.76 DM8 14.56±0.25 3.84±0.17 193.72±6.54 56.72±2.41 32.83±1.66 11.03±0.48 197.11±9.37 46.40±1.57 556.21±22.35 DM9 22.60±0.65 33.44±0.95 72.24±1.98 270.50±8.07 49.54±1.07 5.49±0.07 36.62±0.86 123.99±3.08 614.41±16.57 DM10 9.06±0.20 2.09±0.06 134.33±8.01 38.70±2.11 22.95±1.02 12.46±0.65 155.33±8.08 37.94±1.81 412.86±21.63 DM11 12.67±0.51 2.55±0.13 177.88±4.03 40.91±1.00 28.88±0.96 11.49±0.29 182.83±6.24 35.34±0.81 492.54±13.81 注: “-”表示未鉴定出。下同。
Notes: “—” : Not identified. The same below.Table 2. Content of main anthocyanins in hybrids of C. azalea and C. japonica 'Meili'
μg·g−1 杜鹃红山茶主要花青苷含量为735.81 μg·g−1,山茶‘媚丽’主要花青苷含量为526.61 μg·g−1,杜鹃红山茶主要花青苷含量为‘媚丽’的1.40倍;杂交后代中主要花青苷含量高于杜鹃红山茶的占11.43%,低于山茶‘媚丽’的占37.14%,双亲之间的占51.43%。杂交后代中Cy3GX、Cy3GaX、Cy3GEpCX和Cy3GaEpCX均来源于杜鹃红山茶,其中Cy3GX、Cy3GaX含量低于杜鹃红山茶,而Cy3GEpCX、Cy3GaEpCX含量大部分高于杜鹃红山茶;Cy3G、Cy3Ga、Cy3GEpC和Cy3GaEpC 主要来源于山茶‘媚丽’,含量大部分介于双亲之间。山茶‘媚丽’中含葡萄糖苷的花青苷含量远高于相应含半乳糖苷的花青苷;杜鹃红山茶及杂交后代中含2-O-β-木糖基的花青苷含量基本上高于相应不含2-O-β-木糖基的花青苷,含葡萄糖苷的花青苷(除个别杂交后代外)含量也高于相应含半乳糖苷的花青苷。
-
杜鹃红山茶与山茶‘媚丽’杂交后代主要花青苷比例见表3。杜鹃红山茶花瓣中主要花青苷Cy3GX、Cy3GaX分别占63.14%、19.17%,合计82.30%;其次为Cy3G、Cy3GEpCX和Cy3GaEpCX,分别占6.08%、5.42%和3.17%。山茶‘媚丽’主要成分Cy3GEpC、Cy3G分别占44.76%、40.26%,合计85.02%;其次为Cy3GaEpC、Cy3Ga,分别占8.18%、5.27%。杂交后代中,Cy3GX、Cy3GaX所占比例均低于杜鹃红山茶,而Cy3GaEpCX(除MD72外)所占比例均高于杜鹃红山茶;Cy3G、Cy3Ga、Cy3GEpC和Cy3GaEpC所占比例大部分介于双亲之间,其中,Cy3GX、Cy3GEpCX平均比例分别为31.24%、22.18%,合计53.42%;Cy3G、Cy3GEpC平均比例分别为15.37%和14.20%,合计29.57%。可见,杜鹃红山茶花青苷主要成分为Cy3GX和Cy3GaX,山茶‘媚丽’为Cy3GEpC和Cy3G,杂交后代主要成分为Cy3GX和Cy3GEpCX,其次为Cy3G和Cy3GEpC。
样品Samples Cy3GaX Cy3Ga Cy3GX Cy3G Cy3GaEpC Cy3GaEpCX Cy3GEpCX Cy3GEpC M — 5.27 — 40.26 8.18 — — 44.76 D 19.17 0.78 63.14 6.08 0.28 3.17 5.42 1.61 MD2 2.24 0.45 32.12 6.87 5.61 6.49 33.53 9.10 MD4 0.75 0.92 11.56 22.06 1.42 7.78 14.68 39.73 MD6 1.25 2.06 13.35 39.47 0.47 5.52 9.62 27.11 MD10 4.00 0.35 53.64 4.07 2.76 4.50 23.37 4.77 MD11 4.90 1.50 51.12 14.77 1.53 4.39 16.17 4.35 MD13 10.98 1.60 46.05 7.27 1.50 10.80 14.48 4.88 MD14 1.54 0.99 15.93 11.95 1.76 10.20 16.68 35.98 MD16 4.86 1.73 24.89 11.29 2.49 15.49 23.36 13.28 MD17 1.21 0.86 11.90 12.11 2.73 12.55 14.74 40.47 MD19 2.05 0.69 38.79 15.00 3.53 4.64 21.64 10.84 MD25 9.92 3.47 41.11 20.04 1.32 7.47 8.65 5.56 MD26 3.85 1.27 29.82 22.27 1.20 6.52 14.90 18.68 MD28 2.08 3.31 14.48 49.74 0.71 5.21 6.51 16.59 MD29 13.46 1.85 44.36 9.33 1.99 9.07 14.26 3.33 MD36 3.32 0.66 38.69 7.93 2.27 6.62 31.91 5.46 MD51 5.91 2.04 37.17 11.98 2.73 9.11 21.16 6.79 MD52 9.44 2.90 30.21 14.15 3.13 9.02 17.72 8.98 MD55 2.37 1.60 27.49 29.62 1.75 6.29 11.87 17.03 MD58 0.88 0.22 24.14 7.84 0.88 4.16 37.72 21.69 MD61 2.38 0.80 33.53 11.76 3.25 7.36 25.51 12.22 MD71 2.43 0.17 47.88 3.83 4.25 4.05 31.52 3.79 MD72 3.11 0.37 59.82 10.73 2.54 2.64 16.01 3.41 MD73 1.36 1.18 13.30 19.38 2.05 10.56 18.00 31.51 MD75 2.56 2.32 23.23 34.87 1.27 5.97 9.98 17.62 MD76 0.79 0.42 17.09 18.38 2.01 6.99 20.57 31.81 MD77 15.33 0.98 51.13 5.33 1.40 9.85 12.15 2.23 MD78 0.94 — 28.29 5.34 8.00 4.97 37.54 12.39 DM3 2.41 0.79 29.63 13.19 2.62 5.56 32.49 11.75 DM4 1.77 0.59 20.91 7.06 5.31 8.22 36.60 15.84 DM5 1.96 0.50 32.18 11.00 2.31 5.19 35.26 9.97 DM7 3.24 0.77 36.62 9.14 2.49 6.58 33.78 5.83 DM8 2.57 0.68 34.19 10.01 1.95 5.79 34.77 8.19 DM9 3.59 5.31 11.47 42.95 0.87 7.87 5.82 19.69 DM10 2.15 0.50 31.83 9.17 2.95 5.44 36.82 8.99 DM11 2.53 0.51 35.53 8.17 2.29 5.77 36.50 7.06 Table 3. Proportion of main anthocyanins in hybrids of C. azalea and C. japonica 'Meili'
% -
植物花色是多种因子协同作用的结果,细胞内特定色素的存在是根本内因[33-35]。山茶花的花瓣中色素主要为类黄酮,其中,黄色系山茶花主要为黄酮醇类[36-37];红色系山茶花主要为矢车菊素类[2]。本试验应用高效液相色谱-光电二极管阵列检测器和超高效液相色谱-四极杆-飞行时间质谱联用技术,从杜鹃红山茶及其杂交后代中鉴定出14种花青苷,7种不含2-O-β-木糖基的花青苷与山茶花研究结果相符[2-4],7种含2-O-β-木糖基的花青苷与滇山茶品种‘大理茶’研究一致[11];8种组分含量较高,即Cy3GX、Cy3GaX、Cy3GEpCX和Cy3GaEpCX以及相应不含2-O-β-木糖基的Cy3G、Cy3Ga、Cy3GEpC和Cy3GaEpC;山茶‘媚丽’中未检测到含2-O-β-木糖基花青苷,表明杂交后代中含2-O-β-木糖基花青苷主要遗传自杜鹃红山茶。
本试验中杜鹃红山茶8种主要花青苷总量高于山茶‘媚丽’,杂交后代主要花青苷总量大部分介于双亲之间,少量高于杜鹃红山茶或低于山茶‘媚丽’,表明其杂交后代花青苷总量存在较大的变异。杂交后代中Cy3GX、Cy3GaX含量均低于杜鹃红山茶,Cy3G、Cy3Ga、Cy3GEpC和Cy3GaEpC含量基本上介于双亲之间;但Cy3GEpCX、Cy3GaEpCX含量基本上高于杜鹃红山茶,这可能由于存在杂种优势,具体原因有待于进一步研究。杜鹃红山茶及其杂交后代中,含葡萄糖苷花青苷高于相应含半乳糖苷花青苷,相关结果与已有山茶花青苷研究一致[2-4];含2-O-β-木糖基花青苷含量高于相应不含2-O-β-木糖基花青苷,与滇山茶品种研究结果相符[11,38],表明含2-O-β-木糖基花青苷遗传能力大于相应不含2-O-β-木糖基花青苷。
杜鹃红山茶主要花青苷为Cy3GX和Cy3GaX,二者合计占其花青苷总量的82%以上;山茶‘媚丽’主要为Cy3GEpC和Cy3G,合计占85%以上;杂交后代中主要花青苷Cy3GX和Cy3GEpCX占50%以上,其次是Cy3G和Cy3GEpC约占30%,含2-O-β-木糖基的花青苷所占比例远高于相应不含2-O-β-木糖基的花青苷,也表明含2-O-β-木糖基的花青苷具较强的遗传能力。杂交后代中Cy3GX、Cy3GaX比例降低,Cy3GEpCX、Cy3GaEpCX比例升高,具体原因有待于进一步研究。林艳等[39]对仙客来(Cyclamen persicum Mill)、李晓丽等[40]对月季(Rosa chinensis Jacq.)和陈和明等[41]对秋石斛兰(Dendrobium spp.)的研究结果表明,白色花与红色花杂交后得到的绝大部分是红色花,红色的花色遗传能力强于白色;本试验中杜鹃红山茶鲜红色,山茶‘媚丽’玫瑰红色带白边,其杂交后代大部分花色为红色,少量带白边或零星白斑,也表明其红色遗传能力强于白色。李辛雷等[2-4]研究表明,山茶花的花青苷成分含量与比例均影响其花色,本试验中杜鹃红山茶杂交后代花青苷成分含量与比例对花色的影响不明显,具体原因有待于进一步研究。
本试验中11.43%的杂交后代主要花青苷总量高于杜鹃红山茶,最高为其近2倍(1 406.15 μg·g−1),表现出明显的杂种优势,其花瓣中花青苷具有开发利用价值。通过利用花青苷含量高的杂交后代与杜鹃红山茶回交,有望增加其花青苷的积累,进一步选育出高花青苷含量的新品种。此外,杜鹃红山茶杂交后代具有四季开花的特性,为其花青苷利用提供了稳定的材料,具有良好的开发前景。
-
本试验应用高效液相色谱-光电二极管阵列检测器和超高效液相色谱-四极杆-飞行时间质谱联用技术,对杜鹃红山茶、山茶‘媚丽’及其杂交后代中花青苷进行分析,从杜鹃红山茶及其杂交后代中鉴定出14种花青苷,7种含2-O-β-木糖基的花青苷来源于杜鹃红山茶。杜鹃红山茶主要花青苷总量高于山茶‘媚丽’,杂交后代花青苷总量存在较大的变异,Cy3GX、Cy3GaX含量均低于杜鹃红山茶,Cy3GEpCX、Cy3GaEpCX含量总体上高于杜鹃红山茶,Cy3G、Cy3Ga、Cy3GEpC和Cy3GaEpC含量基本上介于双亲之间。杜鹃红山茶及其杂交后代中含2-O-β-木糖基的花青苷含量高于相应不含2-O-β-木糖基花青苷,含葡萄糖苷花青苷高于相应含半乳糖苷花青苷。杜鹃红山茶主要花青苷为Cy3GX和Cy3GaX,山茶‘媚丽’为Cy3GEpC和Cy3G,杂交后代主要为Cy3GX和Cy3GEpCX,其次为Cy3G和Cy3GEpC,含2-O-β-木糖基的花青苷所占比例高于相应不含2-O-β-木糖基的花青苷,表明含2-O-β-木糖基的花青苷具较强的遗传能力。
Variation Characteristics of Anthocyanin in Hybrids between Camellia azalea and Camellia japonica 'Meili'
- Received Date: 2021-11-26
- Accepted Date: 2022-01-24
- Available Online: 2022-08-20
Abstract:





 DownLoad:
DownLoad: