-
植物的生长和形态具备很强的可塑性,在其生长过程中会形成新的组织,以应对季节和环境的变化[1]。次生维管束是多年生木本植物周期性活动的产物,由维管形成层不断分裂分化形成,其活性随季节更替而变化[2-3]。次生维管组织承担了树木体内物质长距离输导的功能,内含2条主要的长距离输导路径:第一条为木质部,将根系吸收的水分和养分输送到叶片,用于维持蒸腾和光合作用;第二条为韧皮部,将呼吸和生长所需的信号分子和光合产物输送到发育组织和贮藏组织[4]。
温带树木的维管组织在春季复苏,冬季休眠[5]。维管组织在不同时期的形态及生理特性不同[6]。白杨(Populus tomenosa Carr.)活跃期的维管形成层细胞壁中含有较高的甲酯化果胶和较低的酸性果胶,休眠期的维管形成层细胞壁具有较高的酸性果胶,形成层活跃期与休眠期的细胞壁结构也存在很大差异[7]。新生管胞从维管形成层产生后经体积增大,细胞壁加厚,最终发育为成熟管胞[8]。受维管形成层形态和季节的影响,不同时期形成的成熟管胞形态特征如直径、长度及细胞壁厚度等存在明显差异。管胞形态的差异直接影响了管胞的输导能力[9],如冷杉(Abies fabri(Mast.)Craib)的早材输导率约为晚材的11倍,超过90%的输导量来自于早材[10]。同时,树木韧皮部与木质部的内含物及组成成分,如韧皮部的内含多糖颗粒数量、木质部纤维素、木质素、脂类提取物和碳水化合物等,其分布与含量在不同组织和活动期也有显著差异[11]。如云杉(Picea asperata Mast.)的心材比边材含有更多的木质素和更少的纤维素,晚材的半乳糖、葡聚糖和甘露聚糖含量明显高于早材,晚材的果胶含量与亲脂性萃取物较早材更少[12]。辐射松(Pinus radiata D. Don)心材内晚材比早材含有更多的提取物,树脂酸含量也更高[13]。杉木(Cunninghamia lanceolata (Lamb.) Hook.)代谢产物在复苏前后不同,丙酮酸和抗坏血酸含量在复苏后增加[14],并且杉木的早材与晚材的果胶多糖、木聚糖、纤维素和木质素含量差异明显[15]。
马尾松(P. massoniana Lamb.)是一种喜光、不耐荫的速生树种[16],广泛分布于我国南方,是南方面积最大、储量最多的针叶树种之一[17]。目前,有关于马尾松维管组织的发育研究鲜有报道,细胞发育观察也多为马尾松产脂结构的发育研究[18-20],而马尾松次生维管束发育、细胞结构和成分变化仍有待研究。次生维管组织是树木的主体,承担了物质的远距离输导与机械支撑功能。研究维管组织有利于深入揭示树木的次生发育过程。本文通过组织化学分析和细胞离析等手段,研究马尾松纵向输导组织在不同组织的形态、结构及成分变化,旨在揭示维管组织发育过程中细胞的变化特征,为其他树种的维管组织细胞发育研究提供借鉴与参考。
-
采集地位于贵州省花溪区(106°39′ N,26°26′ E),为20世纪80年代飞播造林形成的马尾松林。该区为亚热带高原季风湿润气候,温润多雨,年均气温15.3 ℃,土壤以石灰土为主,为喀斯特地貌区。在林内选取5颗胸径30 cm以上,健康的马尾松成年树,于2022年6月,用凿子于马尾松茎干1.2 ~ 1.5 m处取马尾松木质部与韧皮部材料,取样大小为2 cm × 2 cm × 1 cm,将其裁成0.5 cm × 2 cm × 1 cm后放入FAA固定保存,固定时间在3 d以上,用于韧皮部与木质部的细胞离析。用生长锥于茎干1.2 ~ 1.5 m处钻取木芯,要求韧皮部、形成层与木质部连接完整,随后放入FAA固定保存,固定时间在3 d以上,用于木质部细胞离析。
-
将所采集木芯样本分2组,各5个样本,第一组将修样后长度保留0.5 ~ 1 cm,要求包含完整维管形成层及木质部;第二组修样后保留2021年与2022年完整木质部。将修样后的试验材料经软化,70%、85%、95%、100%乙醇梯度脱水,二甲苯透明,石蜡浸蜡,包埋后于石蜡切片机(Leica RM2235)切片,切片厚度12 μm,经展片,粘片后放入烘箱中40 ℃烘干备用。切片以番红固绿染色,中性树胶封片,显微镜观察,LAS X成像系统拍照。第二组样本中每个样本各选取3列包含完整2021年管胞的细胞列,以Image J统计2021年的马尾松木质部管胞的弦向壁厚度、径向壁厚度、弦向腔径(弦向细胞腔直径)、径向腔径(径向细胞腔直径)、弦向胞径(弦向细胞直径)和径向胞径(径向细胞直径)。同一样本中不同细胞列的细胞层数会存在较小差异,导致不同重复的数据量不同。为便于数据统计,以细胞层数最少的细胞列为标准,将长细胞列多余数据分别在早晚材细胞列的中值周围随机剔除,并求取均值。
-
将裁好的韧皮部材料从固定液中取出,切除周皮。切片观察结果显示,不具输导功能韧皮部约与木质部相距400 ~ 500 μm,因此,在距木质部约500 μm的厚度处将韧皮部切成2块,以富兰克林离析法分别对2块材料进行离析,分别得到2个区域的筛胞。根据木质部的年轮,将木质部的早材和晚材分开,将早晚材转换期管胞刮除干净,每个样本分别切取一部分进行混合离析,得到混合样本的早材管胞和晚材管胞。显微镜观察,DS-Fil成像系统拍照,从离析的早材和晚材管胞中随机选取25个管胞以Image J分别统计管胞上具缘纹孔的纹孔口直径与纹孔缘直径。
-
2组切片经二甲苯脱蜡,无水乙醇浸泡3 min,95%乙醇浸泡3 min后,放入间苯三酚染液中浸染3 min,将浓盐酸滴于切片上,可见木质素的显色反应,同时依据酚类物质的自发荧光现象,结合荧光显微镜(LEICA DM3000)观察,DS-Fil成像系统拍照。纤维素检测选用钙荧光白(CFW)荧光染色试剂盒(GENMED GMS 16034.1),吸取5 μL GENMED染色液至离心管中,加入95 μL蒸馏水将其稀释20倍后得到制备的染色液。将包含早晚材与维管形成层的切片经二甲苯脱蜡,梯度乙醇逐级复水后直接将染色液于暗环境下滴于切片上,室温下静置5 min后以移液枪将染色液吸出,随后吸取GENMED清理液(Reagent A)滴于切片上,用移液枪反复吸取混匀,5 min后吸出,重复2次。最后直接置于荧光显微镜(LEICA DM 3000)下观察。以DS-Fil成像系统拍照,Image J量化第二组5张切片在早晚材相接处木质素与纤维素的组织化学显色和荧光面积。
-
使用excel、SPSS和Origin 2022进行数据处理。依据数据的分布图像,判断管胞层与管胞各系数间为非线性关系,选用非线性模型进行拟合。通过对比origin非线性拟合函数,最后选取了拟合效果较好的Gaussmod函数对不同管胞层的细胞壁厚度、细胞直径与细胞腔径进行拟合。GaussMod函数:
$ y={y}_{0} + \frac{A}{{t}_{0}}{\mathrm{e}}^{\frac{1}{2}{\left(\frac{w}{{t}_{0}}\right)}^{2}-\frac{x-{x}_{c}}{{t}_{0}}}{\int }_{-\infty }^{\frac{x-{x}_{c}}{w}-\frac{w}{{t}_{0}}}\frac{1}{\sqrt{2\Pi }}{{\rm{e}}}^{-\frac{{y}^{2}}{2}}{d}_{y} $
式中:y为管胞的各项测量数据;x为细胞层;y0为模型对x轴的偏移量;w为模型宽度参数;xc为函数峰值对应的细胞层;A为模型面积;t0为辅助参数。
-
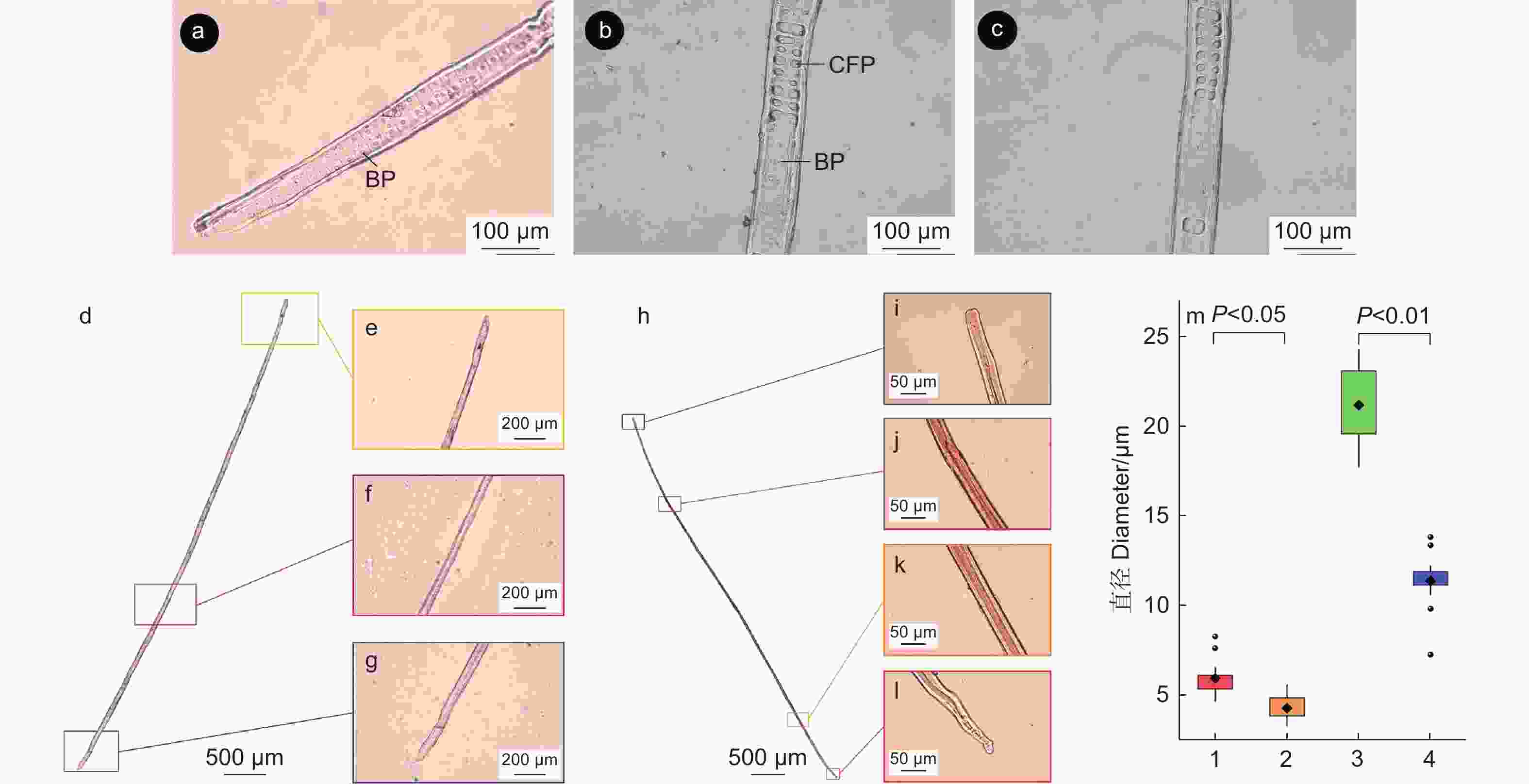
马尾松次生韧皮部主要由筛胞、韧皮薄壁细胞和韧皮射线组成(图1a)。次生韧皮部在发育过程中薄壁组织增大,筛胞会被挤压变形,失去输导功能(图1a)。木质素组织化学和自发荧光观察结果显示,不具输导功能韧皮部出现明显的显色反应(图1b),具输导功能韧皮部与不具输导功能韧皮部出现明显的自发荧光差异(图1c),说明筛胞由具输导功能韧皮部向不具输导功能韧皮部发育的过程中细胞壁发生了木质素沉积。为观察筛胞的结构及不具输导功能韧皮部内筛胞的形变情况,分别离析得到具输导功能韧皮部和不具输导功能韧皮部的完整筛胞,发现马尾松筛胞壁上具筛域,筛域内具筛孔,筛胞由中间向两端渐细(图1d),不具输导功能韧皮部内的筛胞形变严重,胞腔变小,长度低于未形变的筛胞(图1e)。
-
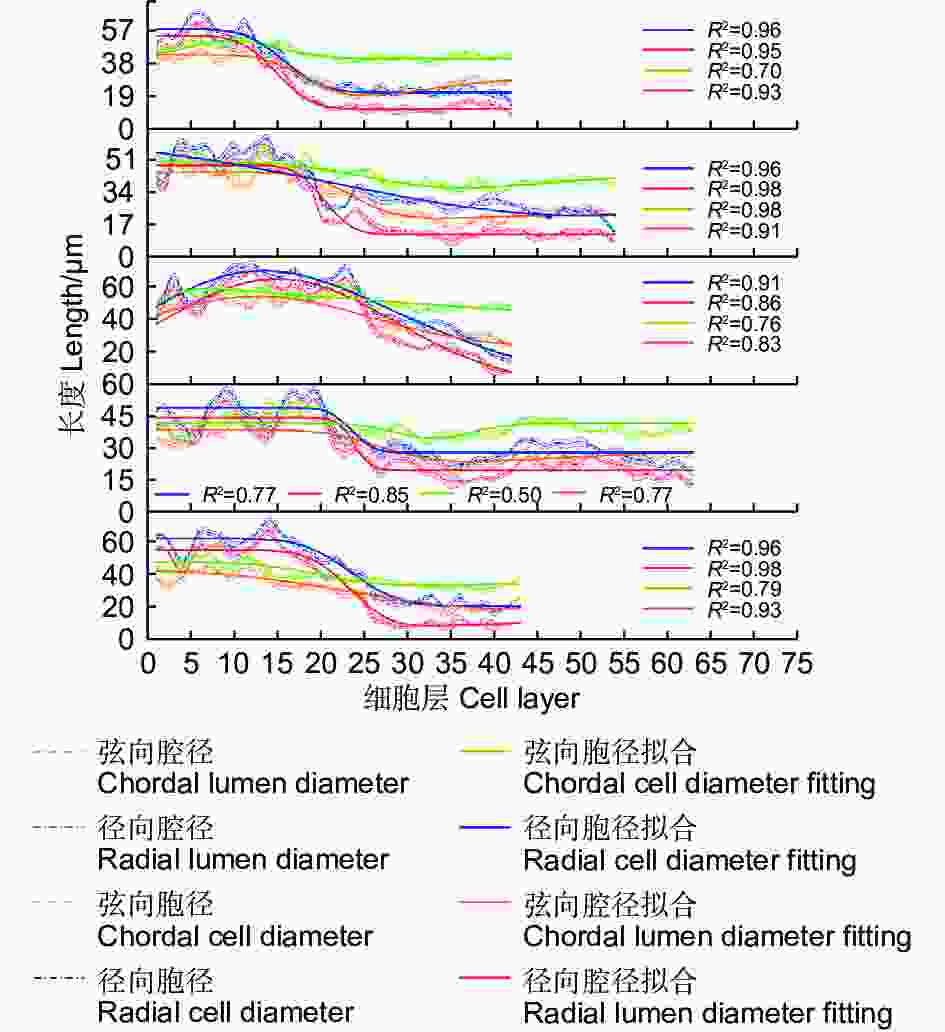
依据细胞形态将木质部划分为早材和晚材,早材和晚材间会出现一段管胞壁逐渐增厚,胞腔逐渐缩小的转换期(图2a),不同样本的管胞壁厚测量结果表明,早材的弦向壁厚及纵向壁厚大部分小于4 μm,而与早材相邻的晚材管胞壁厚大部分大于4 μm,因此,以4 μm为标准划分早材和晚材。
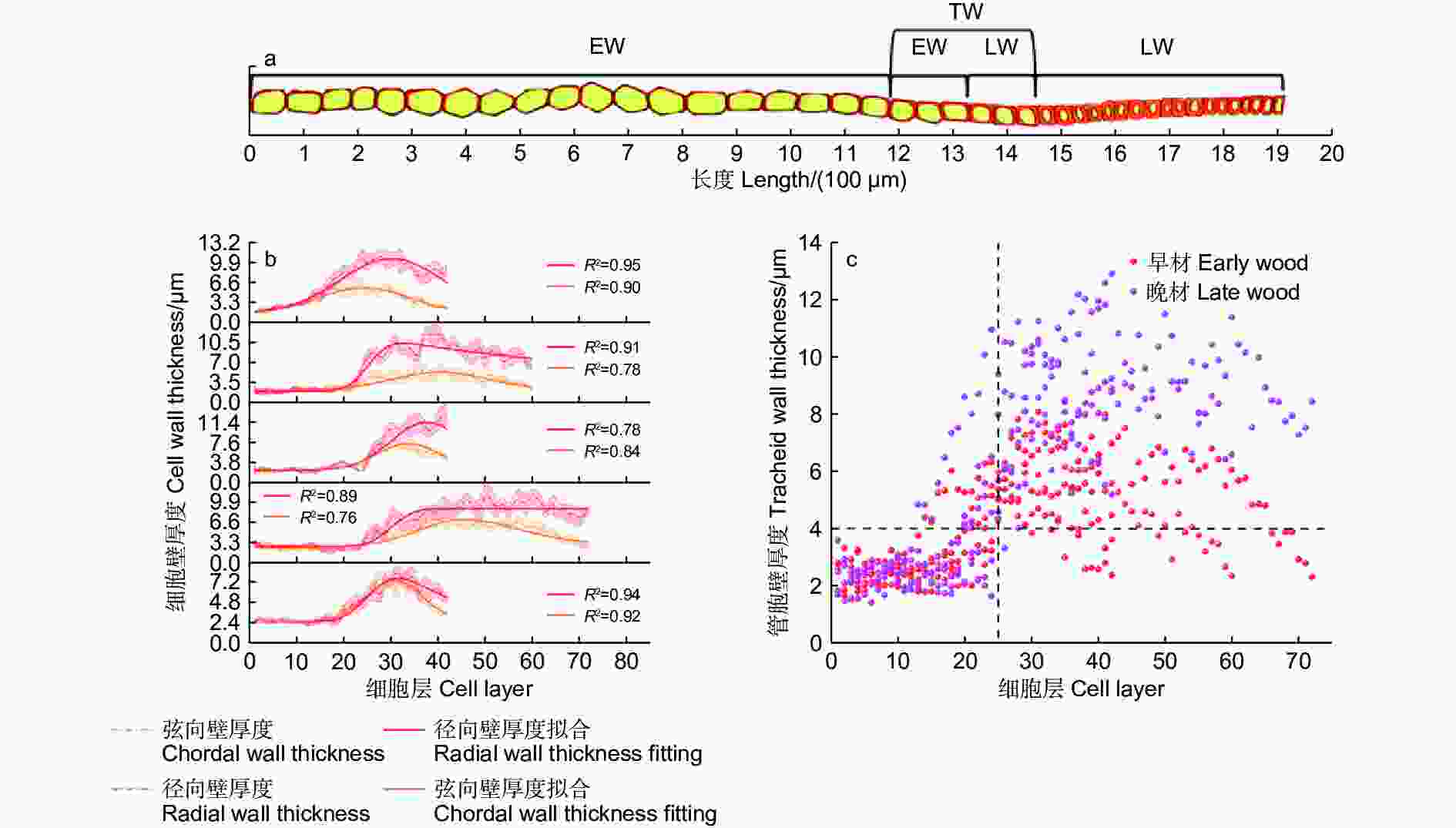
图 2 次生木质部不同样本的管胞壁厚变化
Figure 2. Changes of tracheid wall thickness in different samples of secondary xylem
不同样本的管胞壁厚度、细胞层数和早晚材细胞比例均有差异。管胞弦向壁厚和径向壁厚随管胞层数增加均表现为先增加后降低的变化规律(图2b)。早材管胞的弦向壁厚度与径向壁厚度相近,管胞径向壁在早晚材转换期的曲线斜率与峰值相较于弦向壁更高;到发育后期,弦向壁厚度会逐渐回落至略高于早材的壁厚水平,径向壁厚虽有下降,但仍保持在远高于早材管胞壁厚的水平。
不同样本整合散点图(图2c)表明:马尾松早材主要分布于25层管胞之前,早材管胞壁厚度主要分布于1.5 ~ 3 μm,晚材的管胞层数总体明显高于早材管胞层数,说明成熟林1 a内的晚材管胞形成量高于早材管胞形成量。
不同样本的管胞直径数值存在较大差异,但总体变化趋势相似。管胞腔径与胞径的拟合曲线一致(图3)表明:腔径(d)与胞径(D)随管胞的发育总体呈逐渐降低的趋势;在管胞的发育初期,腔径与胞径的长度表现为D径>D弦,d径>d弦,胞径和腔径变化的拟合曲线斜率相对平稳;当木质部发育进入早晚材的转换期,胞径与腔径拟合曲线斜率降低,其中,kD弦>kD径,kD弦>kd弦,kd弦>kd径,径向胞径和径向腔径在转换期间逐渐低于弦向胞径和弦向腔径;当木质部发育进入晚材期,拟合曲线的斜率再次回升至较平稳的状态,腔径与胞径的长度表现为D弦>D径,d弦>d径。木质部1 a内早晚材的弦向胞径变化较小,变化曲线的斜率相对平稳;而早晚材的弦向腔径、径向胞径和径向腔径的变化较大。
-
木质部细胞离析观察到马尾松管胞有交叉纹孔场与具缘纹孔(图4 a、b)。在管胞的端部(图4 a)和近端部(图4 b)有大量具缘纹孔,纹孔数量从管胞两端至管胞中段逐渐减少,管胞中段几乎见不到具缘纹孔分布(图4 c)。
对木质部的早材和晚材分别进行了细胞离析,分别得到早材和晚材的管胞(图4 d~l)。通过对比观察可知,早材管胞壁上附着的具缘纹孔数量明显多于晚材部分,早材和晚材的纹孔大小也不同。比较分析(图4 m)可知:早材纹孔口与早材纹孔缘的直径显著大于晚材部分,表明木质部由早材发育至晚材的过程中,管胞纹孔数量减少,纹孔口直径和纹孔缘直径也降低。
-
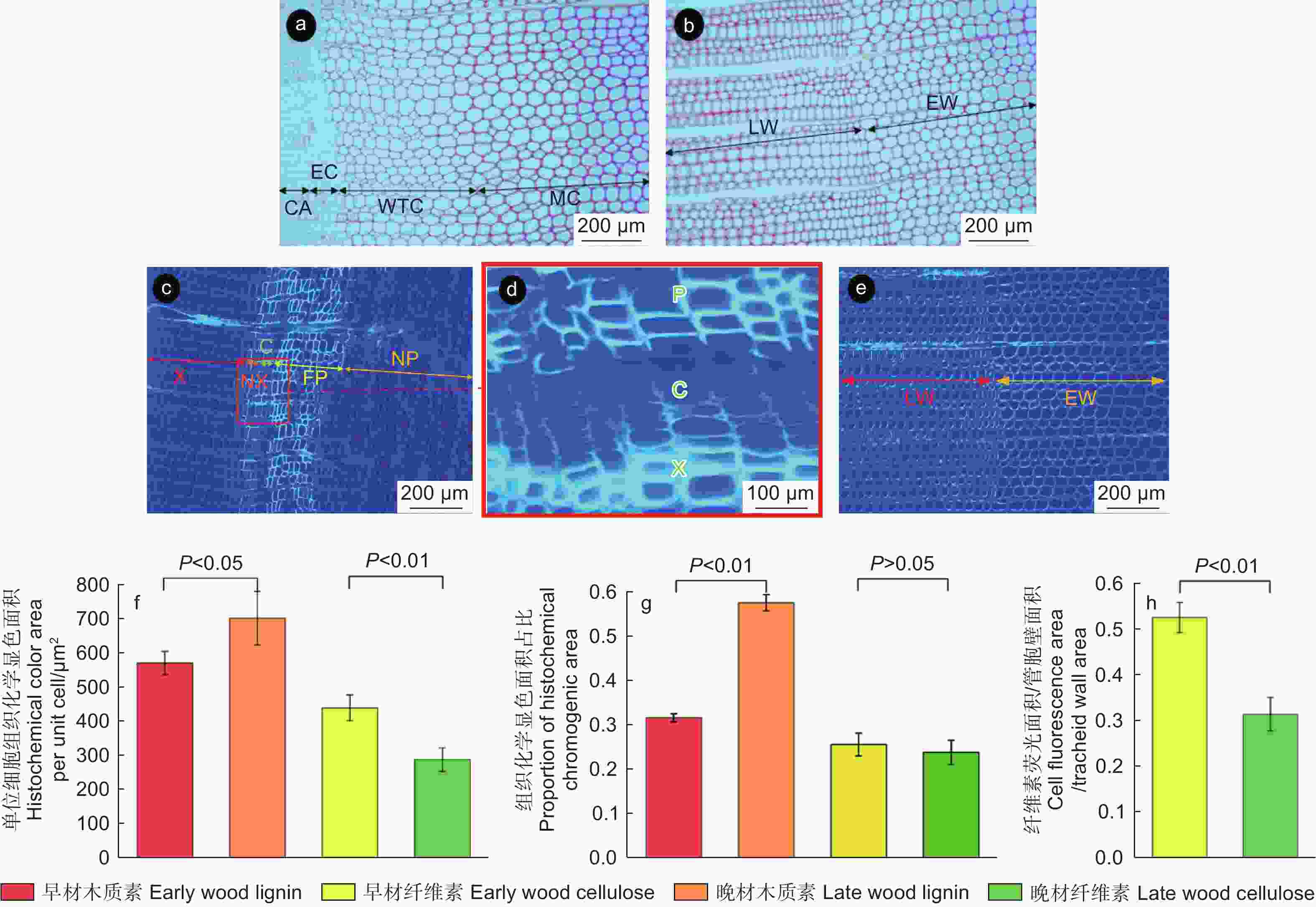
以间苯三酚对发育期的木质部进行木质素组织化学定位,可将木质部发育期的管胞划分为3个时期,分别为径向伸展期、次生壁增厚期和成熟期(图5a)。管胞由维管形成层分化产生后进入径向伸展期,初生壁发育,胞径和胞腔持续增大。当径向伸展期结束后初生壁停止发育,随后管胞进入次生壁增厚期,管胞次生壁持续增厚,次生壁沉积的木质素增多;此后,管胞次生壁停止发育,进入成熟期,发育为成熟管胞。早晚材的木质素组织化学定位显示(图5b),晚材管胞的次生壁厚明显高于早材,而晚材管胞的直径明显小于早材,晚材木质素的分布明显多于早材。
钙荧光白染色后的荧光结果(图5c、d)显示:维管形成层的荧光强度低于新生韧皮部与新生木质部,这表明维管形成层产生新生细胞后经纤维素的富集形成新的管胞与筛胞。木质部的荧光主要集中在新生管胞,韧皮部的荧光主要集中在具输导功能区域,这是由于管胞与筛胞发生木质素沉积后使纤维素的成分占比下降,导致荧光强度降低。早晚材交界处的钙荧光白显色结果(图5e)表明:早材荧光显色较晚材更明显; Image J量化分析结果(图5f~h)表明:晚材木质素显色面积占比和单位细胞显色面积均显著高于早材,表明晚材木质素沉积程度高于早材,而早材纤维素的单位细胞荧光面积和纤维素荧光面积与管胞壁面积的比值极显著高于晚材,表明早材纤维素含量占比高于晚材。
-
树木维管组织的季节性发育伴随着输导功能与机械功能的强弱变化。在韧皮部的研究中,常以筛胞形态变化划分具输导功能韧皮部和不具输导功能韧皮部[21]。本文除观察筛胞形变特征外,还发现马尾松筛胞发育过程中会发生木质素沉积,纤维素占比下降。木质素沉积可增强细胞壁的机械强度,筛胞失去输导能力后机械强度增加,有利于保护韧皮薄壁组织,增加不具输导功能韧皮部的稳定性。
细胞次生壁不仅能增强植物体机械强度,在植物生长、胞间通讯、水分运输和机体防御等方面也发挥重要作用[22]。本研究发现,管胞弦向壁与径向壁厚度随管胞层数增加表现为先大幅上升,后小幅下降的变化趋势,表明早材向晚材变化过程中木质部的机械强度上升。同时,管胞过度加固会降低弹性模量,使水力输导效率下降[23]。管胞径向壁厚度在晚材阶段显著大于弦向壁,而弦向壁厚度在晚材末期会降低至近似于早材的水平。由于相邻管胞的纹孔成对存在,相对而生,共同构成纹孔对[24],晚材末期形成管胞的弦向壁厚度可能会尽量与早材相近,以形成相近的纹孔结构。裸子植物管胞直径与腔径决定了管胞的输导能力[25-26]。管胞的胞径随管胞层数变化递减,导致胞腔逐渐减小,表明管胞的胞内输导能力逐渐降低。
具缘纹孔是管胞间物质输导的通道[24],具缘纹孔直径越大,纹孔导度越大[27]。本文发现,早材的纹孔数量明显多于晚材,早材纹孔缘和纹孔口直径也显著大于晚材部分,表明早材的胞间输导能力显著高于晚材。
木质部的径向伸展期、次生壁增厚期和成熟期是木质部研究中常用的划分方式[28-29]。本文发现,木质部的径向伸展期发生纤维素富集,随后在次生壁增厚期逐渐沉积木质素,并且纤维素占比下降。同时,组织染色表明,晚材木质素沉积程度显著高于早材,纤维素的成分占比下降。Guo对油松(Pinus tabuliformis Carr.)木质部内木质素含量变化的监测结果也表明,由形成早材的活跃期向晚材为主的休眠期转变的过程中,木质素含量持续升高[11]。木质素含量提高还可增强树体的抗逆性 [30],表明管胞由早材向晚材的发育过程是次生木质部为了适应季节变化,抗逆性增强的体现。
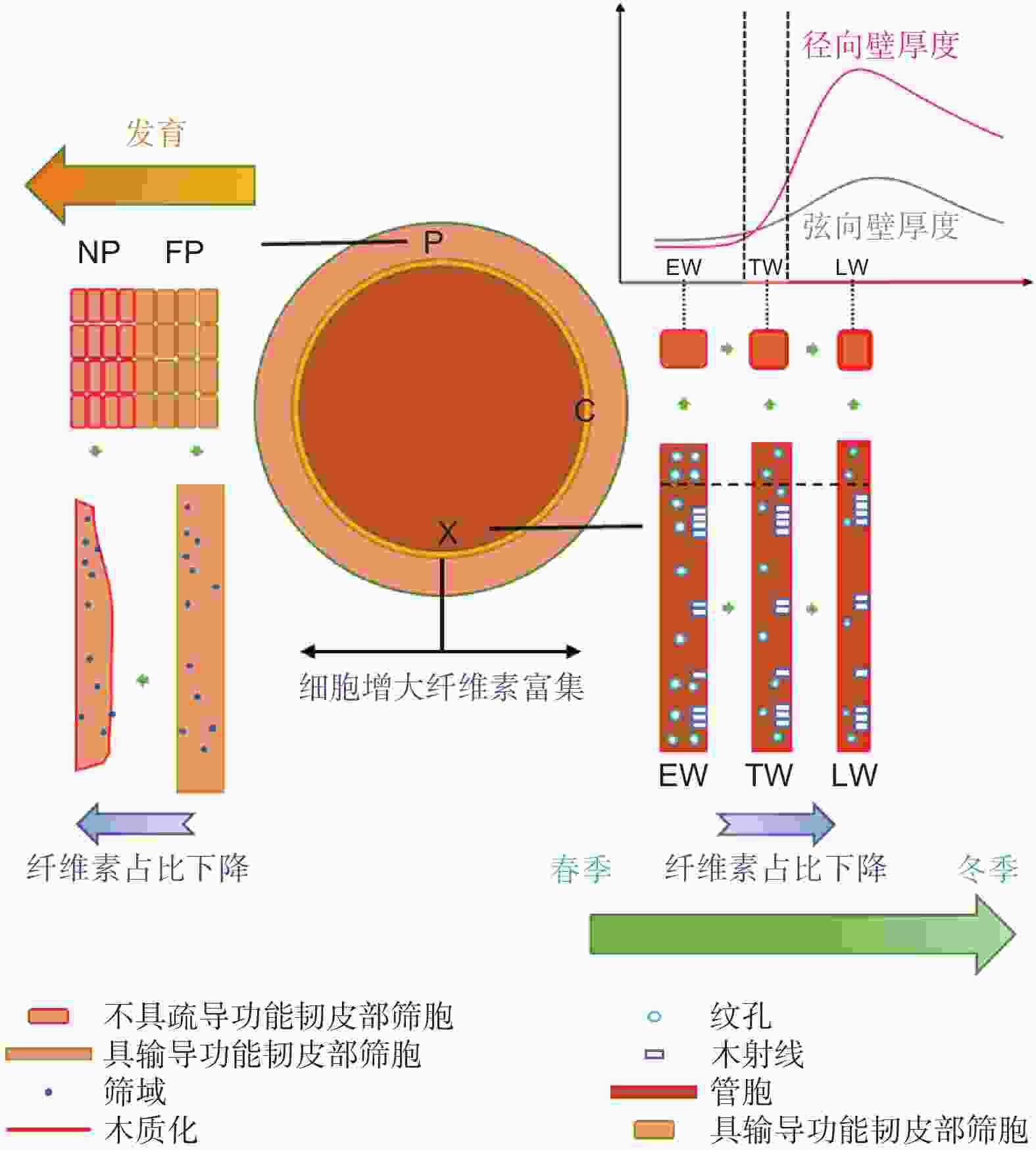
为了揭示马尾松次生维管组织的变化特征,本课题组绘制了马尾松次生维管组织的变化示意图(图6)。维管形成层分化形成新生韧皮部和木质部时会逐渐富集纤维素。木质部管胞发育主要表现为以下几个特征:(1)纹孔直径降低和纹孔数量下降;(2) 次生壁木质素含量增加;(3) 管胞面积下降,主要通过降低细胞径向直径实现;(4)纤维素含量占比下降;(5) 弦向壁和径向壁增厚,但在晚材末期为适应来年早材的纹孔结构和细胞壁厚度会降低至早材水平。韧皮部的发育特征则表现为:(1) 筛胞形变;(2) 筛胞木质化;(3) 纤维素含量占比下降。
-
马尾松维管组织在其发育过程中细胞结构、成分和功能均会发生规律性变化。维管形成层分化形成新生韧皮部和木质部时会逐渐富集纤维素。木质部管胞由早材向晚材发育的过程中,其纹孔直径降低,纹孔数量下降,次生壁木质化程度增加,胞腔面积下降,纤维素含量占比下降,弦向壁及径向壁增厚,表明其胞间及胞内的输导能力逐渐下降,机械强度上升,但在晚材的末期为适应来年早材的纹孔结构和细胞壁厚度会降低至早材水平。韧皮部的发育则表现为筛胞的形变、木质化与纤维素含量占比下降,表明其输导功能丧失和机械强度上升。
马尾松次生维管组织的变化特征
Variation Characteristics of Secondary Vascular Bundles in Pinus massoniana
-
摘要:
目的 旨在揭示马尾松次生维管组织在发育过程中细胞结构、成分和功能的变化规律。 方法 通过组织化学分析和细胞离析等手段,研究马尾松维管组织的细胞形态、结构、木质素和纤维素的变化。 结果 维管形成层分化形成新生韧皮部和木质部时会逐渐富集纤维素;韧皮部发育过程中筛胞发生形变、细胞壁木质化,韧皮部纤维素含量占比下降。木质部管胞由早材向晚材的发育过程中,纹孔直径降低,纹孔数量下降,次生壁木质化程度增加,胞腔面积下降,纤维素含量占比下降,弦向壁及径向壁增厚。 结论 次生维管组织内木质部与韧皮部的细胞结构和成分随发育进程会发生规律性变化,其中,木质部细胞的胞内及胞间输导能力下降,机械强度上升;次生韧皮部丧失输导能力,机械强度上升。 Abstract:Objective This study is to reveal the changes of cellular structure, composition and function of secondary vascular tissue in Pinus massoniana during development. Methods The change of morphology, structure, lignin and cellulose in the vascular tissue of P. massoniana were studied by histochemical analysis and cell segregation. Results The vascular cambium gradually enriched cellulose when it differentiated into new phloem and xylem. During the development of phloem, the sieve cells were deformed and lignified, and the proportion of cellulose in phloem decreased. During the development of xylem tracheids from early wood to late wood, the diameter of striated pores decreased, the number of striated pores decreased, the lignification degree of secondary wall increased, the cell lumen area decreased, the proportion of cellulose content decreased, and the choroidal and radial walls thickened. Conclusion The cellular composition and structure of secondary vascular tissues change regularly with development. The intracellular and intercellular transport capacity of xylem cells decrease, while the mechanical strength increases. The secondary phloem loses its ability to transport and its mechanical strength increases. -
Key words:
- Pinus massoniana
- / secondary vascular bundle
- / structural change
- / histochemical analysis
-
-
[1] GREB T, LOHMANN J U. Plant stem cells[J]. Current Biology, 2016, 26(17): R816-R821. doi: 10.1016/j.cub.2016.07.070 [2] WANG G F, GAO Y F, YANG L W, et al. Identification and analysis of differentially expressed genes in differentiating xylem of Chinese fir (Cunninghamia lanceolata) by suppression subtractive hybridization[J]. Genome, 2007, 50(12): 1141-1155. doi: 10.1139/G07-091 [3] CARVALHO A, PAIVA J, LOUZADA J, et al. The transcriptomics of secondary growth and wood formation in conifers[J]. Molecular Biology International, 2013, 2013: 74324. [4] PETIT G, CRIVELLARO A. Comparative axial widening of phloem and xylem conduits in small woody plants[J]. Trees, 2014, 28(3): 915-921. doi: 10.1007/s00468-014-1006-1 [5] SCHMID R. Review: [Untitled]: Esau's Plant Anatomy: Meristems, Cells, and Tissues of the Plant Body: Their Structure, Function, and Development[J]. Taxon, 2008, 57(2): 678. [6] 许会敏, 王 莉, 曹德昌, 等. 维管形成层活动周期调控研究进展[J]. 科学通报, 2015, 60(7):619-629. [7] CHEN H M, HAN J J, CUI K M, et al. Modification of cambial cell wall architecture during cambium periodicity in Populus tomentosa Carr[J]. Trees, 2010, 24(3): 533-540. doi: 10.1007/s00468-010-0424-y [8] ROSSI S, DESLAURIERS A, ANFODILLO T, et al. Age-dependent xylogenesis in timberline conifers[J]. The New Phytologist, 2008, 177(1): 199-208. doi: 10.1111/j.1469-8137.2007.02235.x [9] HACKE U G, JACOBSEN A L, PRATT R B. Xylem function of arid-land shrubs from California, USA: an ecological and evolutionary analysis[J]. Plant, Cell & Environment, 2009, 32(10): 1324-1333. [10] DOMEC J C, GARTNER B L. How do water transport and water storage differ in coniferous earlywood and latewood?[J]. Journal of Experimental Botany, 2002, 53(379): 2369-2379. doi: 10.1093/jxb/erf100 [11] GUO Y Y, XU H M, WU H Y, et al. Seasonal changes in cambium activity from active to dormant stage affect the formation of secondary xylem in Pinus tabulaeformis Carr[J]. Tree Physiology, 2021, 42(3): 585-599. [12] LLOYD J A. Distribution of extractives in Pinus radiata earlywood and latewood[J]. New Zealand Journal of Forestry Science, 1978, 8(2): 288-294. [13] BERTAUD F, HOLMBOM B. Chemical composition of earlywood and latewood in Norway spruce heartwood, sapwood and transition zone wood[J]. Wood Science and Technology, 2004, 38(4): 245-256. [14] SHI J T, XIA C Y, PENG J Y, et al. Cellular and Metabolite Changes in the Secondary Phloem of Chinese Fir (Cuninghamia lanceolata (Lamb. ) Hook. ) during Dormancy Release[J]. Forests, 2021, 12(11): 1552-1552. doi: 10.3390/f12111552 [15] HONG Y, WU H M, XU H Y, et al. Seasonal development of cambial activity in relation to xylem formation in Chinese fir[J]. Journal of Plant Physiology, 2016, 195: 23-30. doi: 10.1016/j.jplph.2015.12.013 [16] XUE L, HAGIHARA A. Growth analysis on the competition–density effect in Chinese fir (Cunninghamia lanceolata) and Masson pine (Pinus massoniana) stands[J]. Forest Ecology and Management, 2001, 150(3): 331-337. doi: 10.1016/S0378-1127(00)00583-1 [17] XUE L, LIE G, LU G, et al. Allometric scaling among tree components in Pinus massoniana stands with different sites[J]. Ecological Research, 2013, 28(2): 327-333. doi: 10.1007/s11284-012-1021-x [18] 骆 晶, 文晓鹏, 李正春, 等. 马尾松高低产脂种质树脂道解剖结构比较[J]. 西南大学学报(自然科学版), 2020, 42(6):31-37. doi: 10.13718/j.cnki.xdzk.2020.06.004 [19] LI A, WANG Y, WU H. Initiation and development of resin ducts in the major organs of Pinus massoniana[J]. Frontiers of Forestry in China, 2009, 4(4): 501-507. doi: 10.1007/s11461-009-0061-z [20] 耿世磊, 吴玉荷, 赵 晟, 等. 马尾松茎初生树脂道发生和发育研究[J]. 华南农业大学学报, 2000,21(3):44-47. doi: 10.3969/j.issn.1001-411X.2000.03.012 [21] 赵 猛, 亢 晶. 漆树科4种植物次生韧皮部的解剖比较[J]. 林业科学, 2019, 55(6):167-175. doi: 10.11707/j.1001-7488.20190620 [22] COSGROVE DANIEL J. Growth of the plant cell wall[J]. Nature Reviews. Molecular Cell Biology, 2005, 6(11): 850-861. doi: 10.1038/nrm1746 [23] ROSNER S, KARLSSON B. Hydraulic efficiency compromises compression strength perpendicular to the grain in Norway spruce trunkwood[J]. Trees, 2011, 25(2): 289-299. doi: 10.1007/s00468-010-0505-y [24] ZHAO Q, DIXON R A. Transcriptional networks for lignin biosynthesis: more complex than we thought?[J]. Trends in Plant Science, 2011, 16(4): 227-233. doi: 10.1016/j.tplants.2010.12.005 [25] GIBSON A C, CALKIN H W, NOBEL P S. Hydraulic conductance and xylem structure in tracheid-bearing plants[J]. IAWA Journal, 1985, 6(4): 293-302. doi: 10.1163/22941932-90000957 [26] GIBSON A C, CALKIN H W, NOBEL P S. Xylem anatomy, water flow, and hydraulic conductance in the fern cyrtomium falcatum[J]. American Journal of Botany, 1984, 71(4): 564-574. doi: 10.1002/j.1537-2197.1984.tb12542.x [27] DOMEC J C, MEINZER F C, LACHENBRUCH B, et al. Dynamic variation in sapwood specific conductivity in six woody species[J]. Tree Physiology, 2007, 27(10): 1389-1400. doi: 10.1093/treephys/27.10.1389 [28] ČUFAR K, CHERUBINI M, GRIČAR J, et al. Xylem and phloem formation in chestnut (Castanea sativa Mill. ) during the 2008 growing season[J]. Dendrochronologia, 2011, 29(3): 127-134. doi: 10.1016/j.dendro.2011.01.006 [29] PARK J H, CHOI E B, PARK H C, et al. Intra-annual dynamics of cambial and xylem phenology in subalpine conifers at Deogyusan National Park in the Republic of Korea[J]. Journal of Wood Science, 2021, 67(1): 1-10. doi: 10.1186/s10086-020-01935-7 [30] KAACK L, ALTANER C M, CARMESIN C, et al. Function and three-dimensional structure of intervessel pit membranes in angiosperms: a review[J]. IAWA Journal, 2019, 40(4): 673-702. doi: 10.1163/22941932-40190259 -




 下载:
下载: