-
O3作为一种强氧化性的空气污染物,会对农作物和树木造成一定的伤害[1-3]。近地层O3浓度升高是由于其前体物(NOx和VOCs)的排放量增加所导致的[4-5]。近年来,虽然O3的峰值浓度已经得到了有效控制,但均值浓度仍然呈逐渐增加的趋势[6-7]。据预测,北半球中纬度的近地层的O3浓度将每年提高0.5%~2.0%[8]。
O3分子主要是通过叶表的气孔进入组织细胞,并发生反应生成活性氧(ROS),包括羟基自由基(·OH)、超氧阴离子(·O2-)和过氧化氢(H2O2)等[9]。O3会引起叶表出现伤害症状,改变植物的生理生化过程,抑制植物的生长[10-12]。研究表明O3浓度升高会降低三叶草的净光合速率(Pn)和气孔导度(Gs),最终导致生物量降低[10]。Calatayud等[13]的研究表明,植物的根生物量和根茎比在O3浓度升高的条件下会降低,这是由于光合作用产物对根系的分配降低所导致的。O3降低Pn的途径有两种:一种是气孔限制,主要通过降低Gs减少CO2的摄入量[14];另一种是非气孔限制,主要通过减少叶绿素(Chl)含量、降低羧化效率以及抑制电子传输,进而减少CO2的同化[15-16]。在非气孔限制为主要因素的情况下,植物通常会启动抗氧化机制,如提高抗坏血酸和总酚的含量[17]。它们是重要的抗氧化物质,可直接或者间接参与清除活性氧的代谢,被证明可以缓解O3对植物造成的伤害[18-19]。作为抗氧化酶系和非酶系(分子)的总水平,植物总抗氧化能力对O3胁迫的响应,往往与植物的敏感性相关[20]。
在北美和欧洲已经有很多树种,例如欧洲山杨(Populus tremula Michx. × Populus alba L. clone INRA 717-1B4)[21]、晚花稠李(Prunus serotina Ehrh.)[22]、美国白蜡(Fraxinus americana L.)[22]、红花槭(Acer rubrum L.)[22]、欧洲山毛榉(Fagus sylvatica)[23]和欧洲山杨(Populus tremula)[24]等,受到O3的危害。在中国,只有少数树种被用来研究对O3浓度升高的响应,且主要是北方树种[25-27]。然而少有关于中国亚热带常绿阔叶树种的研究。本研究的桢楠(Phoebe zhennan S. Lee et F. N. Wei)和闽楠(Phoebe bournei (Hemsl.) Yang),广泛分布于我国亚热带,为我国所特有,也是该区域常用的造林树种,具有较高的经济和生态价值。此外,桢楠和闽楠作为樟科楠属的不同种类植物,种间的生物学特性之间的差异是否会影响其对O3浓度升高的响应,有必要进行研究。本研究的目的是:(1)研究O3浓度升高对光合作用、抗氧化能力和生物量的影响;(2)比较两树种的O3敏感性并分析原因。
-
本试验的研究地点位于江西省吉安市泰和县灌溪镇的中国科学院千烟洲生态网络站(115°03′29.2″ E, 26°44′29.1″ N),具有典型的亚热带季风气候和红壤丘陵地貌。年均温度为17.8℃,年均降水量为1 471.2 mm,年均蒸发量为259.9 mm,年均相对湿度为83%。
本研究采用开顶式气室(OTCs)进行熏气试验。OTC高2 m,底部为边长1 m的正八边形。为减少OTC外部气体对室内气体的影响,顶部设有一个与水平呈60°的收口。距收口40 cm处设置一圆形的挡雨板。OTC框架由不锈钢构成,室壁材质为钢化玻璃。O3由山东省济南市三康电器有限公司生产的臭氧发生器产生,借助离心式鼓风机经过布气系统进入OTC,并充分混匀。试验设置3个O3浓度水平:未过滤大气(NF)、100 nmol·mol-1(E1)、150 nmol·mol-1(E2)处理。每个O3处理设置3个OTC,共计9个。OTC内的O3浓度用美国Themofisher公司生产的i49型臭氧分析仪进行监测。用北京圣业科技发展有限公司生产的SY-9312D型的质量流量计控制流量,配制设定浓度的O3。
供试植物为1年生桢楠和闽楠盆栽幼苗。选取长势较一致的幼苗,于2014年4月移植于直径20 cm,高30 cm的花盆中,栽培基质为试验地点的土壤。2014年6月初将长势相同的幼苗移入OTC内。每种树木幼苗在每个OTC内有15株。熏气于2014年7月1日开始至11月12日结束,每日熏气8 h(北京时间09:00—17:00),阴雨天停止熏气。试验期间保证各个植株充足且等量的水分供应。
-
收获时用自来水把树木幼苗根系冲洗干净,按根、茎和叶分别统计鲜质量。然后把样品放入75℃的恒温干燥箱内烘干,统计干质量。
-
于晴天上午09:00—11:00从每个OTC内随机选取2~3株幼苗,在每株幼苗主干自上而下4~7叶位选取完全展开的叶2片,用Li-6400便携式光合测定气体交换参数。光合有效辐射(PAR)为1 000 μmol·(m2 s)-1,CO2浓度为周围空气的CO2浓度(约为370 μmol·mol-1),空气流量为0.5 L·s-1,叶室温度为周围空气的温度,相对湿度为55%±5%。
-
在叶片水平上,取2 g叶片组织(避开叶脉),用95%乙醇避光提取48 h,于664、648、470 nm处测定吸光度。根据Harmut等[28]的修正公式计算Chl和类胡萝卜素(Car)含量。
-
称取去叶脉叶片0.2 g左右,加入2 mL的70%冰乙醇在避光条件研磨成匀浆,采用FRAP法测定。加入10 μL样品、90 μL水及900 μL预热至37℃的FRAP反应液(10 mmol·L-1 TPTZ、10 mmol·L-1 FeCl3、0.3 mmol·L-1 醋酸钠缓冲液以1:1:10的比例混合)摇匀后放置40 min后于593 nm处测定吸光值,另以101 000 μmol·L-1(NH4)Fe(SO4)2的标准液代替样品作标准曲线。样品的总抗氧化能力以毫摩尔Fe2+每克提取物表示[29]。
-
称取去除叶脉的叶片组织约0.2 g,放入装有液氮的研钵中,用2 mL10%的三氯乙酸研磨成匀浆,10 000 r·min-1 4℃离心20 min,取上清液,根据Gillespie等[30]的方法测定。
-
采用Folin Ciocaiteu’s酚试剂显色法测定[17]。将30 μL样品加入60 μL 10% Folin Ciocaiteu’s试剂和240 μL 700 mmol·L-1的Na2CO3溶液,于空白酶标板中震荡,置于暗处室温反应30 min后于765 nm处测吸光度。以没食子酸作为标准品代替样品作标准曲线,样品中的总酚以没食子酸的含量表示。
-
所有试验数据以OTC为重复单元。使用SPSS16.0进行统计分析。对于气体交换参数,本研究采用重复测量方差分析的方法,用来分析O3、树种、时间以及它们之间的交互作用。在该方法中,O3和树种作为组间变量,时间作为组内变量。对于其他指标(除气体交换参数),采用一般线性模型中的单变量多因素方差分析的方法,用来分析O3、树种以及它们之间的交互作用。在该方法中,O3和树种作为固定因子。均值之间的显著性差异采用LSD的方法分析(α = 0.05)。采用Origin软件作图。
-
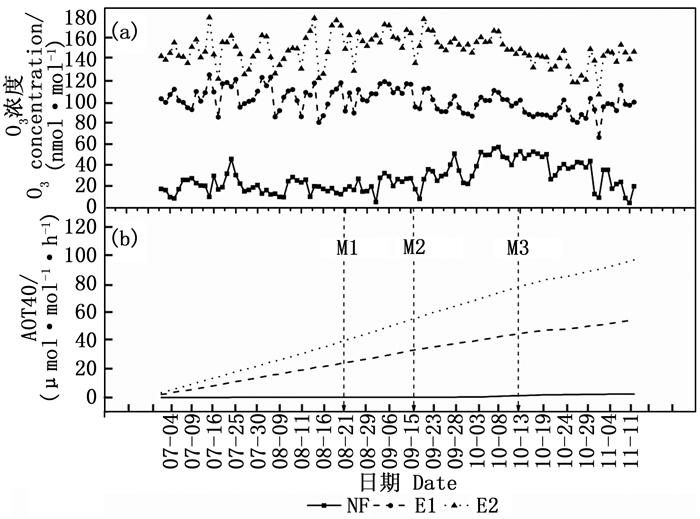
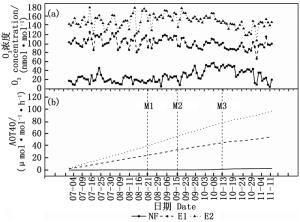
本试验的实际熏气天数为114 d,NF、E1、E2的每日8 h O3浓度均值分别为26.9、99.7、147.0 nmol·mol-1(图 1a)。至试验结束,NF、E1、E2的AOT40(O3浓度超过40 nmol·mol-1的累积)分别为2.4、54.4、96.3 μmol·mol-1·h-1(图 1b)。
-
除树种因素对胞间CO2浓度(Ci)无显著影响外,O3、树种、时间分别对所有测定的气体交换参数有显著影响(P < 0.05)(表 1)。O3×树种×时间对所有测定的气体交换参数有显著影响(P < 0.05)(表 1)。在9月份桢楠的测量中,E1对Pn无显著影响(图 2a)。除此之外,E1和E2显著降低了两树种3次测量的Pn(P < 0.05),且E2的Pn低于E1(除8月份闽楠)(图 2a)。Gs与蒸腾速率(Tr)对O3熏蒸的响应与Pn相似(图 2b和d)。E1显著提高了8月份桢楠的Ci,E2显著提高了8月份桢楠的Ci和9月份闽楠的Ci(P < 0.05)(图 2c)。E2显著降低了10月份桢楠的水分利用效率(WUE),显著降低了8月份和10月份闽楠的WUE(P < 0.05)(图 2e)。E1显著降低了8月份桢楠的气孔限制值(Ls),E2显著降低了8月份和10月份桢楠的Ls(P < 0.05)(图 2f)。
表 1 O3、树种、时间及其交互作用对各种测定参数影响的显著性分析
Table 1. Effects of O3, species, time and their interactions on measured indices
项目
ItemsO3 树种
Species时间
TimeO3×树种
O3×SpeciesO3×时间
O3×Time时间×树种
Time×SpeciesO3×时间×树种
O3×Time×Species净光合速率Pn *** ** *** *** *** *** ** 气孔导度Gs *** ** *** ns ns ns * 胞间CO2浓度Ci ** ns *** * ns ** ** 蒸腾速率Tr *** *** *** ns ns ns ** 水分利用效率WUE *** *** *** ** *** *** ** 气孔限制值Ls ** * *** ** ns *** ** 叶绿素Chl *** *** --- *** --- --- --- 类胡萝卜素Car *** *** --- *** --- --- --- 叶绿素/类胡萝卜素Chl/ Car ** ns --- ns --- --- --- 还原性抗坏血酸Reduced ascorbic acid *** *** --- ns --- --- --- 总酚Total phenols *** *** --- ns --- --- --- 总抗氧化能力Total antioxidant activity ** ns --- ns --- --- --- 叶干质量Foliage biomass *** ** --- * --- --- --- 茎干质量Stem biomass * ns --- ns --- --- --- 根干质量Root biomass *** * --- ns --- --- --- 总干质量Total biomass *** ns --- ns --- --- --- 根茎比Below/above ground biomass ratio ns *** --- * --- --- --- 注:***:在0.001水平下差异显著;**:在0.01水平下差异显著;*:在0.05水平下差异显著;ns:无显著性差;---:无时间因素 Note: ***, **, * means significant difference at 0.001, 0.01 and 0.05; ‘ns’ means no significance; ---means no time factor -
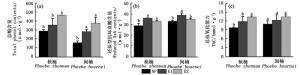
除树种因素对Chl/Car无显著影响外,O3和树种因素分别对Chl、Car含量和Chl/Car具有显著影响(P < 0.01)(表 1)。O3和树种的交互作用对Chl和Car含量具有显著影响(P < 0.001),这说明O3对Car和Chl含量的影响在两树种间具有显著性差异。E1和E2分别显著降低了桢楠的Chl含量23.7%和30.0%(P < 0.05)(图 3a)。E1显著降低了闽楠的Chl含量(P < 0.05)(图 3a)。两树种的Car与Chl含量表现出相似的变化趋势(图 3b)。O3熏蒸略微降低了两树种的Chl/Car,但不显著(图 3c)。
-
除树种因素对总抗氧化能力无显著影响外,O3和树种因素分别对还原型抗坏血酸、总酚和总抗氧化能力水平具有显著影响(P < 0.01)(表 1)。O3和树种因素的交互作用对3种测定的抗氧化物质水平无显著影响(表 1)。O3熏蒸的两树种还原型抗坏血酸、总酚和总抗氧化能力水平高于NF处理(图 4a、b和c)。与NF相比,E1显著提高了桢楠的还原型抗坏血酸含量,显著提高了闽楠的还原型抗坏血酸和总酚含量(P < 0.05)(图 4a、b和c)。E2显著提高了桢楠的3种抗氧化物质水平,显著提高了闽楠的总酚含量(P < 0.05)(图 4a、b和c)。
-
O3因素对所有测定的生物量指标具有显著影响(根茎比除外)(P < 0.05)(表 1)。树种因素对叶干质量、根干质量和根茎比具有显著影响(P < 0.05),但对总干质量和茎干质量的影响不显著(表 1)。O3和树种间的交互作用对叶干质量和根茎比有显著的影响(P < 0.05),说明O3对叶干质量和根茎比的影响在树种间有显著差异(表 1)。除E1对闽楠的茎干质量无显著影响外,E1和E2显著降低了两树种的根、茎、叶和总干质量(P < 0.05),但E1和E2之间无显著差异(图 5a和b)。随着O3浓度升高,闽楠的根茎比显著降低,而桢楠的根茎比变化不显著(图 5c)。
-
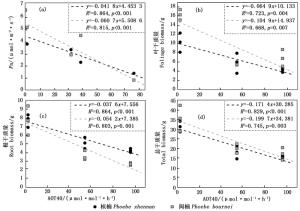
AOT40与两树种的Pn、总干质量、叶、根干质量呈现显著的负相关关系(P < 0.01)(图 6a~d)。闽楠的Pn、总干质量、叶、根干质量减少速率均大于桢楠。闽楠的Pn-AOT40回归方程的斜率较桢楠减少了45.2%(图 6a)。在所有测定的生物量指标-AOT40的回归方程中,闽楠的叶生物量表现出与桢楠的最大差距,较桢楠减少了61.6%(图 6b)。
-
以往的研究表明,高浓度的O3会减少Chl含量并降低Pn[31-32]。对于桢楠,O3浓度越高,Pn和Chl含量的降低程度越大。而对于闽楠,E2的Pn(第一次测定)和Chl含量大于E1,这可能是由于在短期较高浓度的O3熏蒸下,植株通过提高光合能力从而满足自身的正常生长,而E2的后两次测定的Pn小于E1,这说明O3对植株的伤害可能超过了其耐受范围,对Pn造成了更严重的影响。从闽楠的结果可知,Chl含量的变化滞后于Pn,这与Anderson等[14]的研究结果类似。O3熏蒸降低了两树种Pn和Gs的同时,Ls也呈降低趋势,这说明非气孔限制因素是Pn降低的主要因素,这与一些前人的研究结果一致[33-34]。本研究中,Ci增加,WUE减小,说明rubisco相关的限制是CO2同化能力降低的原因,这也映证了Pn的降低主要是由于非气孔限制因素导致的[35-36]。
为了抵御和缓解O3对光合作用的不利影响,植物除了可以在一定程度上关闭气孔从而减少进入叶片的O3通量[37-38],还可以启动抗氧化系统清除活性氧[39-42]。本研究中,O3熏蒸提高了两树种的抗氧化能力,表现为随着O3浓度的升高,总酚含量和总抗氧化能力逐渐升高。然而,两树种E2的还原型抗坏血酸含量低于E1,这可能是由于长期较高浓度的O3熏蒸对抗氧化系统造成了不利影响,这也说明了还原型抗坏血酸较其他抗氧化物质对O3胁迫更加敏感,这与阮亚男等[26]的研究结果相似。O3熏蒸虽然提高了两树种的抗氧化能力,但最终降低了生物量,这说明增加的抗氧化能力不足以抵消O3对植物的伤害。
在本研究中,O3浓度升高一方面降低了两树种的Pn从而减少碳积累,另一方面引起抗氧化物质的合成及其修复过程会消耗干物质。由于这两方面的因素,两树种的生物量最终降低。且Pn降幅较大的树种也是生物量降幅较大的树种,即闽楠,这与Chaudhary等[43]的研究结果相似。O3熏蒸显著降低了闽楠的根茎比,这说明根部受到的影响大于地上部。O3胁迫下根生物量的减少既与CO2同化能力的降低有关,也与从叶分配到根的光合产物的降低有关[44]。而O3熏蒸对桢楠的根茎比无显著影响。
-
在本研究中,两树种的Pn、根、叶、总干质量与AOT40呈显著负相关关系(图 6a~d)。与桢楠相比,闽楠的测定指标(生物量和Pn)与AOT40的线性回归拟合方程的斜率绝对值更大,因此认为闽楠对O3更加敏感。
Zhang等[45]的研究表明,植物的Gs越大,进入叶片组织的O3通量就越大,并表现出更严重的生长负面响应。在本研究中,闽楠的Gs在大多数测量中大于桢楠,较大的Gs和由此产生的较大的O3通量会增加植物的O3敏感性。除此之外,O3熏蒸下桢楠的总抗氧化能力和还原型抗坏血酸含量的增幅大于闽楠。总酚,作为一种有效的自由基清除剂[46],在桢楠中的背景浓度(288.59 μmol·g-1)显著高于闽楠(157.80 μmol·g-1)。综上所述,两树种间的O3敏感性差异与气孔导度和抗氧化物质的背景水平及其对O3浓度升高的响应有关。
-
O3浓度升高主要通过非气孔限制因素降低了两树种的Pn。O3浓度越高,桢楠的Pn越低。而对于闽楠,较高浓度的O3在短期内刺激了Pn的增加,但随着熏气时间的延长,最终对Pn造成了更严重的影响。O3浓度升高虽然提高了两树种的抗氧化能力,但增加的抗氧化能力不足以抵消O3对植物的伤害,最终抑制了生长。与桢楠相比,闽楠的Pn和生物量的降幅更大,因此认为对O3更加敏感。两树种间的O3敏感性差异与气孔导度和抗氧化物质的背景水平及其对O3浓度升高的响应有关。
O3浓度升高对桢楠和闽楠幼苗光合作用、抗氧化能力及生物量的影响
Effects of Elevated O3 Level on Photosynthesis, Antioxidant Capacity and Biomass of Phoebe zhennan and Phoebe bournei Seedlings in Subtropical China
-
摘要:
目的 探讨O3浓度升高对两种亚热带树木幼苗的影响,并分析其敏感性差异及原因。 方法 本试验以1年生桢楠和闽楠幼苗为材料,采用开顶式气室(OTCs),研究未过滤大气、100 nmol·mol-1(E1)、150 nmol·mol-1(E2)O3处理对光合作用、抗氧化能力和生物量的影响。 结果 研究表明:(1)O3熏蒸主要通过非气孔限制因素降低了两树种的净光合速率(Pn)。其中对于桢楠,E2对Pn的不利影响大于E1。而对于闽楠,8月份E2的Pn大于E1,而9月份和10月份小于E1。(2)O3熏蒸提高了两树种的抗氧化能力,表现为总酚含量和总抗氧化能力随着O3浓度的升高逐渐升高,而E2的还原型抗坏血酸含量低于E1。(3)O3熏蒸降低了两树种根、茎、叶及总干质量,及降低了闽楠的根茎比。 结论 O3熏蒸降低了两树种的光合作用,提高了抗氧化能力,最终减少了生物量。根据Pn和生物量,确定闽楠较桢楠对O3浓度升高更加敏感。两树种间的O3敏感性差异与气孔导度和抗氧化物质的背景水平及其对O3浓度升高的响应有关。 Abstract:Objective Considerable researches have documented the negative effects of O3 on woody species in North America and Europe, however, little is known about how woody tree species respond to elevated O3 in subtropical China. The objectives of this study are to investigate the effects of elevated O3 on two evergreen tree species in subtropical China, and compare the O3 sensitivity between them and explore potential mechanisms on differential responses to O3. Method One-year-old seedlings of Phoebe zhennan S. Lee et F. N. Wei and Phoebe bournei (Hemsl.) Yang were exposed to non-filtered air, 100 nmol mol-1 O3 air (E1) and 150 nmol mol-1 O3 air (E2) to investigate the O3 effects on photosynthesis, antioxidant capacity and biomass of these two species. Result The results are as follows:(1) O3 fumigation reduced net photosynthesis rate (Pn) mainly through non-stomatal factors in both species. Pn was lower under E2 than that under E1 in P. zhennan. Whereas for P. bournei, Pn was greater under E2 than that under E1 in August measurement, while the reverse patterns were observed in the measurements in September and October. (2) O3 fumigation increased measured antioxidant substances levels in both species. The levels of total phenols and total antioxidant capacity increased with O3 concentrations increasing. However, reduced ascorbic acid contents were lower under E2 than that under E1. (3) O3 fumigation decreased root, stem, foliage and total biomass in both species. The reduced below/above ground biomass ratio was only observed in P. bournei. Conclusion O3 fumigation decreases photosynthesis, increases the antioxidant capacity, and ultimately inhibits the growth of the two species. The O3 sensibility of P. bournei is stronger than that of P. zhennan based on Pn and biomass. The differential sensitivity to O3 is related to (i) stomatal conductance, and (ii) antioxidant substances responses to elevated O3 and their background level. -
Key words:
- O3
- / photosynthesis
- / antioxidant capacity
- / biomass
- / Phoebe zhennan
- / Phoebe bournei
-
表 1 O3、树种、时间及其交互作用对各种测定参数影响的显著性分析
Table 1. Effects of O3, species, time and their interactions on measured indices
项目
ItemsO3 树种
Species时间
TimeO3×树种
O3×SpeciesO3×时间
O3×Time时间×树种
Time×SpeciesO3×时间×树种
O3×Time×Species净光合速率Pn *** ** *** *** *** *** ** 气孔导度Gs *** ** *** ns ns ns * 胞间CO2浓度Ci ** ns *** * ns ** ** 蒸腾速率Tr *** *** *** ns ns ns ** 水分利用效率WUE *** *** *** ** *** *** ** 气孔限制值Ls ** * *** ** ns *** ** 叶绿素Chl *** *** --- *** --- --- --- 类胡萝卜素Car *** *** --- *** --- --- --- 叶绿素/类胡萝卜素Chl/ Car ** ns --- ns --- --- --- 还原性抗坏血酸Reduced ascorbic acid *** *** --- ns --- --- --- 总酚Total phenols *** *** --- ns --- --- --- 总抗氧化能力Total antioxidant activity ** ns --- ns --- --- --- 叶干质量Foliage biomass *** ** --- * --- --- --- 茎干质量Stem biomass * ns --- ns --- --- --- 根干质量Root biomass *** * --- ns --- --- --- 总干质量Total biomass *** ns --- ns --- --- --- 根茎比Below/above ground biomass ratio ns *** --- * --- --- --- 注:***:在0.001水平下差异显著;**:在0.01水平下差异显著;*:在0.05水平下差异显著;ns:无显著性差;---:无时间因素 Note: ***, **, * means significant difference at 0.001, 0.01 and 0.05; ‘ns’ means no significance; ---means no time factor -
[1] Pretzsch H, Dieler J, Matyssek R, et al. Tree and stand growth of mature Norway spruce and European beech under long-term ozone fumigation[J]. Environmental Pollution, 2010, 158(4):1061-1070. doi: 10.1016/j.envpol.2009.07.035 [2] Sarkar A, Agrawal S. Evaluating the response of two high yielding Indian rice cultivars against ambient and elevated levels of ozone by using open top chambers[J]. Journal of Environmental Management, 2012, 95(2):S19-S24. [3] Wang Y, Yang L, Kobayashi K, et al. Investigations on spikelet formation in hybrid rice as affected by elevated tropospheric ozone concentration in China[J]. Agriculture, Ecosystems & Environment, 2012, 150(1731):63-71. [4] Oltmans S, Lefohn A, Harris J, et al. Long-term changes in tropospheric ozone[J]. Atmospheric Environment, 2006, 40(17):3156-3173. doi: 10.1016/j.atmosenv.2006.01.029 [5] Cape J. Surface ozone concentrations and ecosystem health:past trends and a guide to future projections[J]. Science of the Total Environment, 2008, 400(1):257-269. [6] Derwent R, Simmonds P, Manning A, et al. Trends over a 20-year period from 1987 to 2007 in surface ozone at the atmospheric research station, Mace Head, Ireland[J]. Atmospheric Environment, 2007, 41(39):9091-9098. doi: 10.1016/j.atmosenv.2007.08.008 [7] Dentener F, Keating T, Akimoto H. Hemispheric Transport of Air Pollution 2010: Part A: Ozone and Particulate Matter[R]. Air Pollution Stud, 2011, 17. [8] Vingarzan R. A review of surface ozone background levels and trends[J]. Atmospheric Environment, 2004, 38(21):3431-3442. doi: 10.1016/j.atmosenv.2004.03.030 [9] Yamasaki H, Uefuji H, Sakihama Y. Bleaching of the red anthocyanin induced by superoxide radical[J]. Archives of Biochemistry and Biophysics, 1996, 332(1):183-186. doi: 10.1006/abbi.1996.0331 [10] Chaudhary N, Agrawal S. Intraspecific responses of six Indian clover cultivars under ambient and elevated levels of ozone[J]. Environmental Science and Pollution Research, 2013, 20(8):5318-5329. doi: 10.1007/s11356-013-1517-0 [11] Hoshika Y, Pecori F, Conese I, et al. Effects of a three-year exposure to ambient ozone on biomass allocation in poplar using ethylenediurea[J]. Environmental Pollution, 2013, 180(3):299-303. [12] Watanabe M, Hoshika Y, Koike T. Photosynthetic responses of Monarch birch seedlings to differing timings of free air ozone fumigation[J]. Journal of Plant Research, 2014, 127(2):339-345. doi: 10.1007/s10265-013-0622-y [13] Calatayud V, García-Breijo F J, Cervero J, et al. Physiological, anatomical and biomass partitioning responses to ozone in the Mediterranean endemic plant Lamottea dianae[J]. Ecotoxicol Environ Saf, 2011, 74(5):1131-1138. doi: 10.1016/j.ecoenv.2011.02.023 [14] Anderson P D, Palmer B, Houpis J L, et al. Chloroplastic responses of ponderosa pine (Pinus ponderosa) seedlings to ozone exposure[J]. Environment International, 2003, 29(2):407-413. [15] Kivimäenpää M, Selldén G, Sutinen S. Ozone-induced changes in the chloroplast structure of conifer needles, and their use in ozone diagnostics[J]. Environmental Pollution, 2005, 137(3):466-475. doi: 10.1016/j.envpol.2005.01.033 [16] HUANG S, ZHAO T-h, JIN D-y, et al. Photosynthetic physio-response of urban Quercus mongolica leaves to surface elevated ozone concentration[J]. Liaoning Forestry Science and Technology, 2009(5):1-4. [17] Kähkönen M P, Hopia A I, Vuorela H J, et al. Antioxidant activity of plant extracts containing phenolic compounds[J]. Journal of Agricultural and Food Chemistry, 1999, 47(10):3954-3962. doi: 10.1021/jf990146l [18] Dizengremel P, Le Thiec D, Bagard M, et al. Ozone risk assessment for plants:central role of metabolism-dependent changes in reducing power[J]. Environmental Pollution, 2008, 156(1):11-15. doi: 10.1016/j.envpol.2007.12.024 [19] Yamaji K, Julkunen-Tiitto R, Rousi M, et al. Ozone exposure over two growing seasons alters root-to-shoot ratio and chemical composition of birch (Betula pendula Roth)[J]. Global Change Biology, 2003, 9(10):1363-1377. doi: 10.1046/j.1365-2486.2003.00669.x [20] Zhang W, Feng Z, Wang X, et al. Responses of native broadleaved woody species to elevated ozone in subtropical China[J]. Environmental Pollution, 2012, 163(4):149-157. [21] Bagard M, Le Thiec D, Delacote E, et al. Ozone-induced changes in photosynthesis and photorespiration of hybrid poplar in relation to the developmental stage of the leaves[J]. Physiologia Plantarum, 2008, 134(4):559-574. doi: 10.1111/ppl.2008.134.issue-4 [22] Schaub M, Skelly J, Zhang J, et al. Physiological and foliar symptom response in the crowns of Prunus serotina, Fraxinus americana and Acer rubrum canopy trees to ambient ozone under forest conditions[J]. Environmental Pollution, 2005, 133(3):553-567. doi: 10.1016/j.envpol.2004.06.012 [23] Braun S, Schindler C, Leuzinger S. Use of sap flow measurements to validate stomatal functions for mature beech (Fagus sylvatica) in view of ozone uptake calculations[J]. Environmental Pollution, 2010, 158(9):2954-2963. doi: 10.1016/j.envpol.2010.05.028 [24] Nikula S, Percy K, Oksanen E, et al. Effects of elevated ozone on growth and foliar traits of European and hybrid aspen[J]. Boreal Environment Research, 2009, 14(suppl.A):29-47. [25] 付伟, 高江艳, 徐胜, 等.高浓度臭氧对城市白桦和银中杨光合作用的影响[J].生态学杂志, 2014, 33(12):3184-3190. [26] 阮亚男, 何兴元, 陈玮, 等.臭氧浓度升高对油松抗氧化系统活性的影响[J].应用生态学报, 2009, 20(5):1032-1037. [27] 张巍巍, 赵天宏, 王美玉, 等. O3浓度升高对油松光合作用的影响[J].农业环境科学学报, 2007, 26(3):1024-1028. doi: 10.3321/j.issn:1672-2043.2007.03.043 [28] Harmut A, Lichtenthaler K. Chlorophylls and carotenoids:pigments of photosynthetic membranes[J]. Method Enzymol, 1987, 148(34):350-383. [29] Benzie I, Strain J. Ferric reducing/antioxidant power assay:direct measure of total antioxidant activity of biological fluids and modified version for simultaneous measurement of total antioxidant power and ascorbic acid concentration[J]. Methods in Enzymology, 1998, 299(1):15-27. [30] Gillespie K M, Ainsworth E A. Measurement of reduced, oxidized and total ascorbate content in plants[J]. Nature Protocols, 2007, 2(4):871-874. doi: 10.1038/nprot.2007.101 [31] Elvira S, Alonso R, Castillo F J, et al. On the response of pigments and antioxidants of Pinus halepensis seedlings to Mediterranean climatic factors and long-term ozone exposure[J]. New Phytologist, 1998, 138(3):419-432. doi: 10.1046/j.1469-8137.1998.00136.x [32] Zheng Y, Shimizu H, Barnes J. Limitations to CO2assimilation in ozone-exposed leaves of Plantago major[J]. New Phytologist, 2002, 155(1):67-78. doi: 10.1046/j.1469-8137.2002.00446.x [33] Zheng Y, Lyons T, Ollerenshaw J H, et al. Ascorbate in the leaf apoplast is a factor mediating ozone resistance in Plantago major[J]. Plant Physiology and Biochemistry, 2000, 38(5):403-411. doi: 10.1016/S0981-9428(00)00755-5 [34] Zhang W, Niu J, Wang X, et al. Effects of ozone exposure on growth and photosynthesis of the seedlings of Liriodendron chinense (Hemsl.) Sarg, a native tree species of subtropical China[J]. Photosynthetica, 2011, 49(1):29-36. [35] Mikkelsen T. Physiological responses of Fagus sylvatica L. exposed to low levels of ozone in open-top chambers[J]. Trees, 1995, 9(6):355-361. [36] Reichenauer T G, Bolhàr-Nordenkampf H R. Mechanisms of impairment of the photosynthetic apparatus in intact leaves by ozone[J]. Zeitschrift für Naturforschung C, 1999, 54(9-10):824-829. doi: 10.1515/znc-1999-9-1031 [37] Calatayud A, Iglesias D J, Talón M, et al. Response of Spinach Leaves (Spinacia oleracea L.) to Ozone Measured by Gas Exchange, Chlorophyll a Fluorescence, Antioxidant Systems, and Lipid Peroxidation[J]. Photosynthetica, 2004, 42(1):23-29. [38] Matyssek R, Wieser G, Ceulemans R, et al. Enhanced ozone strongly reduces carbon sink strength of adult beech (Fagus sylvatica)-Resume from the free-air fumigation study at Kranzberg Forest[J]. Environmental Pollution, 2010, 158(8):2527-2532. doi: 10.1016/j.envpol.2010.05.009 [39] Löw M, Herbinger K, Nunn A, et al. Extraordinary drought of 2003 overrules ozone impact on adult beech trees (Fagus sylvatica)[J]. Trees, 2006, 20(5):539-548. doi: 10.1007/s00468-006-0069-z [40] 张巍巍, 牛俊峰, 王效科, 等.大气臭氧浓度增加对湿地松幼苗的影响[J].环境科学, 2011, 32(6):1710-1716. [41] 金明红, 冯宗炜, 张福珠.臭氧对水稻叶片膜脂过氧化和抗氧化系统的影响[J].环境科学, 2000, 21(3):1-5. [42] Yu H, Chen Z, Shang H, et al. Physiological and biochemical responses of Machilus ichangensis Rehd. et Wils and Taxus chinensis (Pilger) Rehd. to elevated O3 in subtropical China[J]. Environmental Science and Pollution Research, 2017, 24(21):17418-17427. doi: 10.1007/s11356-017-9417-3 [43] Chaudhary N, Singh S, Agrawal S, et al. Assessment of six Indian cultivars of mung bean against ozone by using foliar injury index and changes in carbon assimilation, gas exchange, chlorophyll fluorescence and photosynthetic pigments[J]. Environmental Monitoring and Assessment, 2013, 185(9):7793-7807. doi: 10.1007/s10661-013-3136-0 [44] Andersen C P. Source-sink balance and carbon allocation below ground in plants exposed to ozone[J]. New Phytologist, 2003, 157(2):213-228. doi: 10.1046/j.1469-8137.2003.00674.x [45] Zhang J, Ferdinand J, Vanderheyden D, et al. Variation in gas exchange within native plant species of Switzerland and relationships with ozone injury:An open-top experiment[J]. Environmental Pollution, 2001, 113(2):177-185. doi: 10.1016/S0269-7491(00)00175-5 [46] Sarkar A, Rakwal R, Bhushan Agrawal S, et al. Investigating the impact of elevated levels of ozone on tropical wheat using integrated phenotypical, physiological, biochemical, and proteomics approaches[J]. Journal of Proteome Research, 2010, 9(9):4565-4584. doi: 10.1021/pr1002824 -



 下载:
下载: